INTRODUCTION
Coronary artery fistula is not a rare anomaly characterized by abnormal connections between coronary arteries and cardiac chambers or pulmonary vasculatures. It is usually congenital [1], resulting from incomplete obliteration of the primitive myocardial sinusoids and fistulous tracts. On the other hand, acquired coronary artery fistulas are extremely rare and have been reported as complications after chest trauma [2], myocardial infarction [3], myocardial biopsy [4], cardiac surgery [5], percutaneous transluminal coronary angioplasty [6]. However, formation of coronary fistula by guidewire induced trauma occurs very rarely.
We reported a case of an arteriovenous fistula originating from the left circumflex (LCX) artery, draining into the left posterior ventricular vein as a result of guidewire induced trauma.
CASE REPORT
A 51-year-old man presented worsening exertional angina. His other relevant medical history includes hypercholesterolemia, hypertension, and diabetes mellitus. Medications were atenolol 50 mg daily, aspirin 100 mg daily daily, nocorandil 10 mg twice a day.
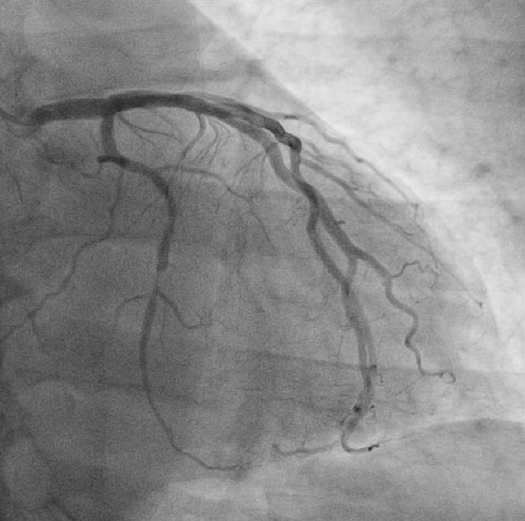
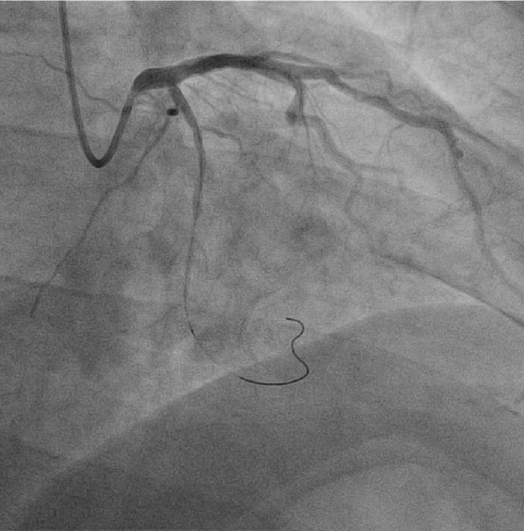
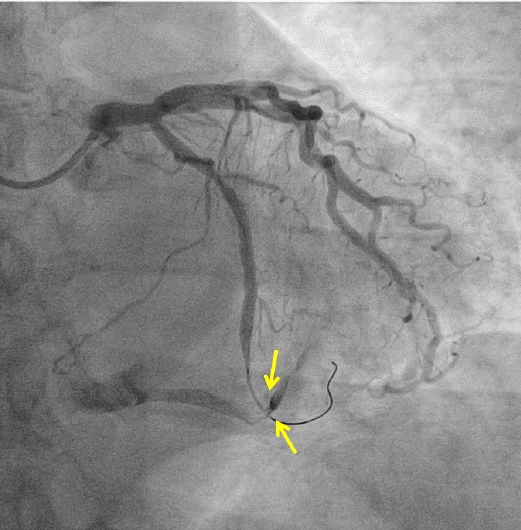
Coronary angiography revealed significant stenosis of distal circumflex artery. The left anterior descending artery and right coronary artery showed no significant luminal narrowing (Fig. 1). Subsequently, PCI was performed after the risks were explained and informed consent obtained. A 6 French arterial sheath was inserted into the left radial artery, and unfractionated heparin (8,000 units) was initially administered. A 0.014 inch Runthrough (Terumo, Tokyo, Japan) guidewire was passed across the distal circumflex stenosis lesion and into the distal vessel (Fig. 2). The distal circumflex coronary lesion was dilated using 2.0 ├Ś 15 mm Splint balloon (Medtronic, Minneapolis, USA) to 10 atmospheres and stented using 3.0 ├Ś 24 mm zotarolimus-eluting coronary stent (Medtronic, Minneapolis, USA) to 16 atmospheres. The result was satisfactory. However, at this stage, a distal perforation resulting in a coronary arteriovenous fistula within the distal circumflex artery was noted (Fig. 3). The contrast at this site cleared with slow resolution indicating distal runoff into the left posterior ventricular vein.
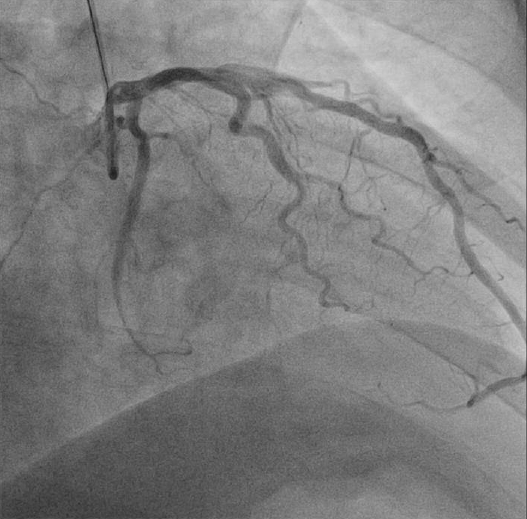
We suspected that guidewire moved to and fro during PCI and wire exit was the cause for perforation and coronary arterioveous fistula. Fortunately, the patientŌĆÖs state was stable. Emergency echocardiogram revealed no pericardial effusion. We observed patient without reversal of anticoagulation with protamin sulphate infusion in the catheterization lab. Coronary angiography revealed resolution of the fistula 20 minites later (Fig. 4). We determined close observation in coronary care unit and maintained antiplatelets medication. Echocardiogram was within normal limits with no effusion or wall-motion abnormalities. He was discharged a few days later.
DISCUSSSION
Iatrogenic coronary artery fistula is an extremely rare complication of percutaneus transluminal angioplasty. The natural history of iatrogenic coronary artery fistula has not been reported and is not known well because limited cases, but reported cases shows variable courses from stable state to rapid detoriation. Probable mechanisms of iatrogenic coronary fistula were sub-intimal balloon inflation [7], guide wire perforation [8], over expansion of a coronary segment and inappropriate wire tracking. The majority of iatrogenic coronary artery fistula caused by percutaneous coronary intervention involve communications between the coronary arteries and the cardiac chambers such as the right ventricle [9], and less commonly, the left ventricle [10].
In present case, an iatrogenic coronary arteriovenous fistula from the distal left circumflex artery to the left posterior ventricular vein as a complication of guidewire induced trauma was occurred. Although the guidewire selection is influenced by criteria related to the vessel anatomy, the lesion morphology, initially floppy wire has been recommended. Free wire tip motions are important to avoid catching the tip on plaque and to prevent vessel trauma, such as dissection ofperforation of the vessel. After acrossing the lesion, it is important to monitor the distal position of the wire tip.
The acute intraprocedural management of a coronary artery fistula resulting from percutanous intervention is not established. A variety of approaches can be used to manage coronary artery fistulas. However, in view of the paucity of case reports describing the present complication, it is difficult to define its optimal management. It would be reasonable, however, to attempt closure of the fistula in the first instance by prolonged balloon inflation at the site of rupture. Emergency surgical closure of the fistula may not be necessary as some close spontaneously, while others remain patent but cause no symptoms. If this fails, and particularly if the patientŌĆÖs condition deteriorates, then surgical closure of the fistula ought to be considered. Recently, Percutaneous treatment has since become the preferred treatment option and can be performed with detachable balloons [11], coil embolization [12], a PTFE-covered stent [13].
In conclusion, cautious guidewire passing to the distal coronary artery, correct matching of balloon to vessel size and caution in negotiating complex lesions should minimize the chances of this complication occurring.



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print






