 |
 |
| Korean J Med > Volume 96(2); 2021 > Article |
|
Abstract
Diabetes is one of the important risk factors in cardiovascular disease associated with atherosclerosis, and cardiovascular disease is the leading cause of death in patients with diabetes mellitus. Recent randomized placebo-controlled cardiovascular outcome trials of all new antidiabetic drugs have linked SGLT-2 inhibitors and GLP1-agonists to not only increased cardiovascular stability but significant reduction of cardiovascular disease. These results have led to preferential selection of the most effective and beneficial antidiabetic drugs with the evidence of cardiovascular safety and efficacy. Herein, we address cardiovascular stability and the effectiveness of antidiabetic drugs, focusing on recently developed ones.
Ùï╣Ùç¿Ùèö ÙÅÖÙºÑÛ▓¢ÝÖöýÖÇ Û┤ÇÙá¿ÙÉ£ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿýØÿ Û░Çý×Ñ ýñæýÜöÝò£ ýØ©ý×É ýñæ ÝòÿÙéÿÙí£, Ùï╣Ùç¿Ù│æýØ┤ ý×êÙèö Û▓¢ýÜ░ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿýØÿ ý£äÝùÿÙÑáýØ┤ 2-3Ù░░ ýØ┤ýâü ýªØÛ░ÇÝò£Ùïñ. ýï¼ÝÿêÛ┤Ç ýºêÝÖÿýØÇ Ùï╣Ùç¿Ù│æ ÝÖÿý×ÉýØÿ ýú╝ýÜö ýé¼ÙºØ ýøÉýØ© ýñæ ÝòÿÙéÿýØ┤Ù®░, Ýè╣Ý×ê ýï¼ÝÿêÛ┤Ç ýºêÝÖÿýØÿ ý£äÝùÿÙÑáýØÇ Ùï╣Ùç¿ýØÿ ý£áÙ│æ Û©░Û░äýùÉ Ùö░ÙØ╝ ýªØÛ░ÇÝòÿÛ│á Û│áÝÿêýòò, ýØ┤ýâüýºÇýºêÝÿêýªØ, ÙîÇýé¼ýªØÝøäÛÁ░, Ùºîýä▒ ýïáý×Ñ ýºêÝÖÿÛ│╝ Û░ÖýØÇ ÙÅÖÙ░ÿ ýºêÝÖÿ ý£áÙ¼┤ýùÉ ýÿüÝûÑýØä Ù░øÙèöÙïñ[1]. Ùö░ÙØ╝ýä£ Ùï╣Ùç¿ ÝÖÿý×ÉýùÉýä£ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿýØÿ ýÿêÙ░®Û│╝ ý╣ÿÙúîÙèö ÙºñýÜ░ ýñæýÜöÝòÿÙïñ. Ùï╣Ùç¿ ý╣ÿÙúîýò¢ýᣠýñæ ÝòÿÙéÿýØ© Ùí£ýï£Û©ÇÙª¼ÝâÇýí┤(rosiglitazone)ýØ┤ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿýØÿ ý£äÝùÿýØä ýªØÛ░Çýï£Ýé¿ÙïñÙèö Û▓âýØ┤ ÝÖòýØ©ÙÉ£ Ýøä Ù»©ÛÁ¡ýïØÝÆêýØÿýò¢ÛÁ¡(Food and Drug Administration, FDA)ýØÇ 2008ÙàäÙÂÇÝä░ Ù¬¿Ùôá Ùï╣Ùç¿ ý╣ÿÙúîýò¢ýá£ýùÉ ÙîÇÝò£ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿ Ù░£ýâØ ý×ÉÙúîÙÑ╝ ýÜöÛÁ¼ÝòÿýÿÇÙïñ[2,3]. ýØ┤ýùÉ Ùö░ÙØ╝ DPP-4 ýûÁýá£ýá£(dipeptidyl peptidase 4 inhibitor, DPP4-inhibitor)ÙÑ╝ ýï£ý×æý£╝Ùí£ ýâêÙí£ýÜ┤ Ùï╣Ùç¿ ý╣ÿÙúîýò¢ýá£Ùôñ Ù¬¿ÙæÉýùÉýä£ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿ Ù░£ýâØýùÉ ÙîÇÝò£ ý×äýâü ýù░ÛÁ¼ÙÑ╝ ÝåÁÝò┤ Ùï╣Ùç¿ ý╣ÿÙúîýò¢ýá£ÙôñýØÿ ýï¼ÝÿêÛ┤Ç ýòêýáòýä▒ýùÉ ÙîÇÝò£ ý×äýâüýáü ÛÀ╝Û▒░Û░Ç ýá£ýï£ÙÉÿýùêÙïñ. ýØ┤Ùƒ¼Ýò£ Û░ÇýÜ┤Ùì░ ýÁ£ÛÀ╝ Û░£Ù░£ÙÉ£ Ùï╣Ùç¿Ù│æ ýò¢ýá£ýØ© SGLT-2 ýûÁýá£ýá£(sodium-glucose cotransporter 2 inhibitor, SGLT2 inhibitor)ýÖÇ GLP-1 ýêÿýÜ®ý▓┤ ý×æýÜ®ýá£(glucagon-like peptide 1 receptor agonist, GLP-1 agonist)Ùèö ýï¼ÝÿêÛ┤Ç ýòêýáòýä▒ýØ┤ ý×êýØä Ù┐É ýòäÙïêÙØ╝ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿýØÿ Ù░£ýâØýØä Û░Éýåîýï£ÝéñÙèö Ùì░ Ù¬àÙ░▒Ýò£ ýØ┤ýáÉýØ┤ ý×êýØîýØ┤ ÝÖòýØ©ÙÉÿýùêÛ│á ýØ┤ÙÑ╝ ÝåáÙîÇÙí£ Û░ÇýØ┤Ùô£ÙØ╝ýØ©ýØÿ Ù│ÇÝÖö Ù░Å ý×äýâüýùÉ ÙºÄýØÇ Ù│ÇÝÖöÛ░Ç ýØ╝ýû┤Ùé¼Ùïñ[4]. Û│╝Û▒░ýùÉ Ùï╣Ùç¿Ù│æ ý╣ÿÙúî Ù¬®Ýæ£Û░Ç ÝÿêÙï╣ýØÿ ýáüýáêÝò£ ýí░ýáêýùÉ ý×êýùêÙïñÙ®┤ ýÁ£ÛÀ╝ýùÉÙèö ýï¼ÝÿêÛ┤Ç ýºêÝÖÿýØÿ ý£äÝùÿÙÅä Û▓¢Û░ÉýùÉ Ùìö ý┤êýáÉýØ┤ Ùº×ýÂ░ýá© ý×êý£╝Ù®░, Ùï╣Ùç¿ ý╣ÿÙúîýò¢ýá£ýØÿ ýäáÝâØýùÉ ý×êýû┤ ýï¼ÝÿêÛ┤Ç ýòêýáòýä▒ Ù░Å ýï¼ÝÿêÛ┤Ç ý£áÝÜ¿ýä▒ýØÿ ýªØÛ▒░Û░Ç ý×êÙèö ýò¢ýá£ÙÑ╝ ýÂöý▓£Ýò┤ýò╝ ÝòÿÙèö ý╣ÿÙúî Ýî¿Ùƒ¼Ùïñý×äýØÿ Ù│ÇÝÖöÙÑ╝ Û░Çýá©ýÿ¿ Û▓âýØ┤Ùïñ. ýØ┤ýùÉ Ùö░ÙØ╝ ýÁ£ÛÀ╝ Ù»©ÛÁ¡Ùï╣Ùç¿Ù│æÝòÖÝÜî(American Diabetes Association, ADA)ýùÉýä£Ùèö ýï¼ÝÿêÛ┤Ç ýºêÝÖÿýØ┤ ý×êÛ▒░Ùéÿ ÙïñýêÿýØÿ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿ ý£äÝùÿýØ©ý×ÉÛ░Ç ý×êÙèö Û▓¢ýÜ░ ýØ┤ 2Û░ÇýºÇ ýò¢ýá£ÙÑ╝ ý▓ÿÙ░®ÝòÿÙØ╝Û│á ÛÂîÛ│áÝòÿÛ│á ý×êÙïñ[1,4]. ýØ┤ýùÉ Ù│©Û│áýùÉýä£Ùèö ýÁ£ÛÀ╝ Û░£Ù░£ÙÉ£ Ùï╣Ùç¿ ý╣ÿÙúî ýò¢ýá£ÙÑ╝ ýñæýï¼ý£╝Ùí£ ýï¼ÝÿêÛ┤Ç ýòêýáòýä▒Û│╝ ÝÜ¿Û│╝ÙÑ╝ ýÜöýò¢ÝòÿÛ│á ý×äýâüýáü ýáüýÜ®ýØä ýåîÛ░£ÝòÿÛ│áý×É Ýò£Ùïñ.
ýÁ£ÛÀ╝ Û░£Ù░£ÙÉ£ Ùï╣Ùç¿ ý╣ÿÙúîýò¢ýá£ýùÉÙèö DPP-4 ýûÁýá£ýá£, GLP-1 ýêÿýÜ®ý▓┤ ý×æýÜ®ýá£, SGLT-2 ýûÁýá£ýá£Û░Ç ý×êÙïñ. Û░ü ýò¢ýá£ýØÿ ýï¼ÝÿêÛ┤Ç ýòêýáòýä▒ýØä ÝÖòýØ©ÝòÿÙèö Ù¼┤ý×æý£ä ý£äýò¢ ÙîÇýí░ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿ Ù░£ýâØ ýù░ÛÁ¼(randomized placebo-controlled CV outcome trials)ýùÉýä£ ýØ╝ý░¿ ýù░ÛÁ¼ ýóàÙºÉýØÇ ýï¼ÝÿêÛ┤ÇÛ│ä Û┤ÇÙá¿ ýé¼ÙºØ, Ù╣äý╣ÿÙ¬àýáü ýï¼ÛÀ╝Û▓¢ýâë ÙÿÉÙèö Ù╣äý╣ÿÙ¬àýáü Ùçîýí©ýñæý£╝Ùí£ ýáòýØÿÙÉÿÙèö 3-point ýú╝ýÜö ýï¼ÝÿêÛ┤ÇÛ│ä Û┤ÇÙá¿ ýé¼Û▒┤(major adverse cardiovascular event, MACE)ý£╝Ùí£ ýáòýØÿÙÉÿýùêÛ│á, ýÂöÛ░Çýáüý£╝Ùí£ ýï¼ÙÂÇýáäýùÉ ýØÿÝò£ ý×àýøÉ, ÙÂêýòêýáòýä▒ Ýÿæýï¼ýªØýùÉ ýØÿÝò£ ý×àýøÉ, ý┤Ø ýé¼ÙºØýØÿ ýóàÝò® Ýÿ╣ýØÇ Û░üÛ░üýØÿ Û▓░Û│╝ÙÑ╝ ÝåÁÝò┤ ýòêýáòýä▒ Ù░Å ÝÜ¿Û│╝ÙÑ╝ ÝÖòýØ©ÝòÿýÿÇÙïñ. ÛÀ© Û▓░Û│╝ ýØ╝ÙÂÇ ýò¢ýᣠýù░ÛÁ¼ýùÉýä£Ùèö ýï¼ÝÿêÛ┤Ç ýòêýáòýä▒ ÝÖòýØ©Û│╝ Ýò¿Û╗ÿ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿ ýÿêÙ░® ÝÜ¿Û│╝Û╣îýºÇ ÝÖòýØ©ýØ┤ ÙÉÿÙ®┤ýä£ ýÂöÛ░ÇýáüýØ© ýù░ÛÁ¼ÙôñýØ┤ ýºäÝûëÙÉÿÛ│á ý×êÙïñ. ýòäÙ×ÿýùÉ Û░ü ýò¢ýá£Ù│äÙí£ ýù░ÛÁ¼ ý×äýâü ýù░ÛÁ¼ Û▓░Û│╝ÙÑ╝ ýä£ýêáÝòÿÛ│á Ýæ£Ùí£ ýáòÙª¼ÝòÿýÿÇÙïñ.
DPP-4 ýûÁýá£ýá£Ùèö ýñæÙô▒ÙÅäýØÿ Ùï╣ Û░òÝÖö ÝÜ¿Û│╝Û░Ç ý×êÛ│á ýáÇÝÿêÙï╣ýØÿ ý£äÝùÿýØ┤ Ùé«ýòä ÙºÄýØÇ ÝÖÿý×ÉýùÉýä£ ýé¼ýÜ®ÙÉÿÙèö Ùï╣Ùç¿ ý╣ÿÙúîýò¢ýᣠýñæ ÝòÿÙéÿýØ┤Ùïñ. DPP-4 ýûÁýá£ýá£Ùèö ýåîý×ÑýùÉýä£ ÙÂäÙ╣äÙÉÿÙèö ýØ©Ýü¼ÙáêÝï┤(incretin)ýØ© glucagon-like-peptide 1 (GLP-1)ýØä ÙÂäÝò┤Ýòÿýù¼ ÙÂêÝÖ£ýä▒ÝÖö ýï£ÝéñÙèö ÝÜ¿ýåîýØ© DPP-4ÙÑ╝ ýûÁýá£ÝòÿÙèö ýù¡ÝòáýØä ÝòÿÙ®░, ýØ┤ÙÑ╝ ÝåÁÝò┤ ýØ©ýèÉÙª░ ÙÂäÙ╣äÙÑ╝ ý┤ëýºäÝòÿÛ│á ýØ©ýèÉÙª░ ý×æýÜ® ýï£Û░äýØä ýªØÛ░Çýï£ÝéñÙ®░ Û©ÇÙú¿ý╣┤Û│ñ ÙÂäÙ╣äÙÑ╝ ýûÁýá£Ýò¿ý£╝Ùí£ýì¿ ÝÿêÙï╣ýØä Û░Éýåîýï£Ýé¿Ùïñ[5]. Û░Çý×Ñ Ù¿╝ýáÇ Ù░£Ýæ£ÙÉ£ Saxagliptin Assessment of vascular Outcomes Recorded in Patients with Diabetes Mellitus-Thrombolysis in Myocardial Infarction 53 (SAVOR-TIMI 53) ýù░ÛÁ¼ýùÉýä£Ùèö 3-point MACEýùÉ ÙîÇÝò┤ saxagliptin ýé¼ýÜ®ÛÁ░Û│╝ ÙîÇýí░ÛÁ░ Û░äýØÿ ý£áýØÿÝò£ ý░¿ýØ┤Û░Ç ýùåýùêÛ│á, ýØ┤Ýøä Ù░£Ýæ£ÙÉ£ alogliptinÛÁ░Û│╝ ÙîÇýí░ÛÁ░ýØä Ù╣äÛÁÉÝò£ Examination of Cardiovascular Outcomes with Alogliptin versus Standard of Care (EXAMINE) ýù░ÛÁ¼ýÖÇ sitagliptinÛÁ░Û│╝ ÙîÇýí░ÛÁ░ýØä Ù╣äÛÁÉÝò£ Trial to Evaluate Cardiovascular Outcomes After Treatment With Sitagliptin (TECOS) ýù░ÛÁ¼, linagliptinÛÁ░Û│╝ ÙîÇýí░ÛÁ░ýØä Ù╣äÛÁÉÝò£ The Cardiovascular and Renal Microvascular Outcome Study With Linagliptin (CARMELINA) ýù░ÛÁ¼ Ù¬¿ÙæÉýùÉýä£ÙÅä ýï¼ÝÿêÛ┤Ç ýºêÝÖÿýùÉ ÙîÇÝò£ ý£áýØÿÝò£ ý░¿ýØ┤ÙÑ╝ Ù│┤ýØ┤ýºÇ ýòèýòÿÙïñ(Table 1) [6-9]. ýØ┤ÙôñýØÇ Ù¬¿ÙæÉ Ù╣äýù┤Ùô▒ýä▒ ý×äýâü ýù░ÛÁ¼Ùí£ýì¿, ýØ┤ ýù░ÛÁ¼ Û▓░Û│╝Ùí£ DPP-4 ýûÁýá£ýá£ýØÿ ýï¼ÝÿêÛ┤Ç ýòêýáòýä▒ýØ┤ ý×àýªØÙÉÿýùêÙïñ. ÙïñÙºî, linagliptinÛ│╝ sitagliptinýùÉýä£Ùèö ý£áýØÿÝòÿýºÇ ýòèýòÿýºÇÙºî SAVOR-TIMI 53 ýù░ÛÁ¼ Û▓░Û│╝ saxagliptinýØ┤ ÙîÇýí░ÛÁ░ýùÉ Ù╣äÝò┤ ýï¼ÙÂÇýáäýùÉ ýØÿÝò£ ý×àýøÉýØä 27%Û░ÇÙƒë ýØÿÙ»© ý×êÛ▓î ýªØÛ░Çýï£ÝéñÙèö Û▓âýØ┤ ÝÖòýØ©ÙÉÿýùêÛ│á, ýØ┤Ýøä DPP-4 ýûÁýá£ýá£ýØÿ ýï¼ÙÂÇýáäýùÉ ýØÿÝò£ ý×àýøÉ ýªØÛ░Ç Ù¼©ýá£Û░Ç ÙîÇÙæÉÙÉÿÙ®┤ýä£ ýºäÝûëÙÉ£ alogliptinýØÿ Ýòÿý£ä ýù░ÛÁ¼ýùÉýä£Ùèö ýï¼ÙÂÇýáäýØä Û©░ýáÇýùÉ ýºäÙï¿Ù░øýòÿÙìÿ ÝÖÿý×ÉýùÉýä£Ùèö alogliptin Ýê¼ýò¢ÛÁ░Û│╝ ÙîÇýí░ÛÁ░ Û░äýùÉ ýï¼ÙÂÇýáäýùÉ ýØÿÝò£ ý×àýøÉýùÉ ÙîÇÝò┤ ý£áýØÿÝò£ Û▓░Û│╝ÙÑ╝ Ù│┤ýØ┤ýºÇ Ù¬╗ÝòÿýÿÇý£╝Ùéÿ, Û©░ýáÇýùÉ ýï¼ÙÂÇýáäýØä ýºäÙï¿Ù░øýºÇ ýòèýòÿÙìÿ ÝÖÿý×ÉýùÉýä£Ùèö alogliptinýØä ýé¼ýÜ®Ýò£ ÛÁ░ýùÉýä£ ÙîÇýí░ÛÁ░ýùÉ Ù╣äÝò┤ ýï¼ÙÂÇýáäý£╝Ùí£ ýØ©Ýò£ ý×àýøÉýØä 76% ýªØÛ░Çýï£ÝéñÙèö Û▓âýØ┤ ÝÖòýØ©ÙÉÿýùêÙïñ[7,10]. Û▓░ÛÁ¡ Ùï╣Ùç¿ ÝÖÿý×ÉýùÉýä£ DPP4 ýûÁýá£ýá£Ùèö ýï¼ÝÿêÛ┤Ç ýòêýáòýä▒ýØÇ ý×àýªØÙÉÿýùêý£╝Ùéÿ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿ ýÿêÙ░®ýùÉ ý£áýØÿÝò£ ÝÜ¿Û│╝Ùèö ýùåý£╝Ù®░, ýØ╝ÙÂÇýùÉýä£Ùèö ýï¼ÙÂÇýáäý£╝Ùí£ ýØ©Ýò£ ý×àýøÉ ýªØÛ░ÇýØÿ ý£äÝùÿýØ┤ ý×êÙïñ.
GLP-1 ýêÿýÜ®ý▓┤ ý×æýÜ®ýá£Ùèö Ýö╝Ýòÿýú╝ýé¼ýᣠÙï╣Ùç¿ ý╣ÿÙúîýò¢ýá£Ùí£ DPP-4 ýûÁýá£ýá£ýÖÇ ý£áýé¼ÝòÿÛ▓î ýØ©Ýü¼ÙáêÝï┤ýØÿ ý×æýÜ®ýØä ýªØÛ░Çýï£ý╝£ ÝÿêÙï╣ Û░òÝòÿ ÝÜ¿Û│╝ÙÑ╝ Ù│┤ýØ©Ùïñ[11]. GLP-1 ýêÿýÜ®ý▓┤ ý×æýÜ®ýá£Ùèö Ùï╣Ùç¿ ý╣ÿÙúîýò¢ýᣠýñæ ýï¼ÝÿêÛ┤Ç ýòêýáòýä▒ýùÉ ÙîÇÝò£ ý×àýªØÛ│╝ Ýò¿Û╗ÿ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿ ýÿêÙ░®ýùÉ ÝÜ¿Û│╝Û░Ç ý×êÙèö ýÁ£ý┤êýØÿ ýò¢ýá£Ùí£ ÙîÇÙæÉÙÉÿýùêÙïñ. GLP-1 ýêÿýÜ®ý▓┤ ý×æýÜ®ýᣠýñæ ÙîÇÝæ£ýáü ýò¢ýá£ýØ© liraglutideýØÿ ý×äýâü ýù░ÛÁ¼(Liraglutide Effect and Action in Diabetes: Evaluation of Cardiovascular Outcome Results, LEADER)ýùÉýä£Ùèö liraglutideýØÿ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿýùÉ ÙîÇÝò┤ ýòêýáäÝòá Ù┐É ýòäÙïêÙØ╝ ý╣ÿÙúîÛÁ░ýØ┤ ÙîÇýí░ÛÁ░Û│╝ Ù╣äÛÁÉÝòÿýù¼ ý£áýØÿÝòÿÛ▓î 3-point MACEÙÑ╝ Û░Éýåîýï£ÝéñÙèö Û▓âýØä ÝÖòýØ©ÝòÿýÿÇÙïñ[12]. ÙÿÉÝò£, ÙïñÙÑ© GLP-1 ýêÿýÜ®ý▓┤ ý×æýÜ®ýá£ýØ© semaglutideÙÑ╝ ýé¼ýÜ®Ýò£ Trial to Evaluate Cardiovascular and Other Long-term Outcomes With Semaglutide in Subjects With Type 2 Diabetes (SUSTAIN-6) ýù░ÛÁ¼, albiglutideýÖÇ ÙîÇýí░ÛÁ░ýØä Ù╣äÛÁÉÝò£ Albiglutide and cardiovascular outcomes in patients with type 2 diabetes and cardiovascular disease (Harmony) ýù░ÛÁ¼ ÛÀ©Ùª¼Û│á dulaglutideýÖÇ ÙîÇýí░ÛÁ░ýØä Ù╣äÛÁÉÝò£ Researching Cardiovascular Events With a Weekly Incretin in Diabetes (REWIND) ýù░ÛÁ¼ýùÉýä£ÙÅä Ù¬¿ÙæÉ ýï¼ÝÿêÛ┤Ç ýòêýáòýä▒Û│╝ Ýò¿Û╗ÿ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿ ýÿêÙ░®ýùÉ Û©ìýáòýáüýØ© Û▓░Û│╝ÙÑ╝ Ù│┤ýÿÇÙïñ(Table 2) [13-15]. ÙÿÉÝò£, GLP-1 ýêÿýÜ®ý▓┤ ý×æýÜ®ýá£Û░Ç ÝÿêÙï╣ ýí░ýáê ýÖ©ýùÉÙÅä ýØ©ýèÉÙª░ ýáÇÝò¡ýä▒ Û░£ýäá, ý▓┤ýñæ Û░Éýåî Ù░Å ýØ┤ýâüýºÇýºêÝÿêýªØýØÿ Û░£ýäá Ùô▒ýØä ÝåÁÝò┤ Ùï╣Ùç¿Ù│æ ý×Éý▓┤ýØÿ Ù░£ýâØ ý£äÝùÿýØä Ùé«ýÂöÛ│á ÙîÇýé¼ýªØÝøäÛÁ░ýØÿ Û░£ýäáýùÉÙÅä ÙÅäýøÇýØä ýú╝Ùèö Û▓âýØ┤ ÝÖòýØ©ÙÉÿýû┤ ýØ┤Ùí£ ýØ©Ýò£ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿ ýÿêÙ░® ÝÜ¿Û│╝ÙÅä ý×êÙèö Û▓âý£╝Ùí£ ýâØÛ░üÙÉ£Ùïñ[16]. ÛÀ©Ùƒ¼Ùéÿ GLP-1 ýêÿýÜ®ý▓┤ ý×æýÜ®ýᣠýñæ lixisenatideýÖÇ exenatideÙÑ╝ ýé¼ýÜ®Ýò£ Evaluation of Lixisenatide in Acute Coronary Syndrome (ELIXA) ýù░ÛÁ¼ Ù░Å Exenatide Study of Cardiovascular Event Lowering (EXSCEL) ýù░ÛÁ¼ýùÉýä£Ùèö ýï¼ÝÿêÛ┤Ç ýòêýáäýä▒ýØÇ Û▓ÇýªØÙÉÿýùêý£╝Ùéÿ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿ ýÿêÙ░® ÝÜ¿Û│╝Ùèö ý×àýªØÝòÿýºÇ Ù¬╗ÝòÿýÿÇÙïñ[17,18]. Ùö░ÙØ╝ýä£ Ýÿäý×¼ ADAýùÉýä£Ùèö Ýè╣Ý×ê ýï¼ÝÿêÛ┤Ç ýºêÝÖÿýØ┤ ý×êÙèö ÝÖÿý×É Ù░Å ÙïñýêÿýØÿ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿ ý£äÝùÿýØ©ý×ÉÛ░Ç ý×êÙèö ÝÖÿý×ÉýØÿ Û▓¢ýÜ░ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿ ýÿêÙ░®ÝÜ¿Û│╝Û░Ç ý×êÙèö 4Û░ÇýºÇ ýò¢ýá£(liraglutide, albiglutide, semaglutide, dulaglutide)ýØÿ Ýê¼ýò¢ýØä ÛÂîÛ│áÝòÿÛ│á ý×êÙïñ[1].
SGLT-2 ýûÁýá£ýá£Ùèö ý¢®ÝîÑýØÿ ÛÀ╝ý£äÙÂÇ ýä©Ùç¿Û┤ÇýùÉýä£ Ùï╣ýØÿ ý×¼ÝØíýêÿÙÑ╝ Ùï┤Ùï╣ÝòÿÙèö SGLT-2 ýêÿýÜ®ý▓┤ÙÑ╝ ýûÁýá£Ýòÿýù¼ ýåîÙ│Çý£╝Ùí£ Ùï╣ Ù░░ýäñýØä ý┤ëýºäýï£ÝéñÛ│á ÛÀ© Û▓░Û│╝ Ýÿêýòí Ùé┤ ÝÿêÙï╣ýØä Û░Éýåîýï£ÝéñÙèö ýò¢ýá£ýØ┤Ùïñ[19]. SGLT-2 ýûÁýá£ýᣠýñæ ýÁ£ý┤êÙí£ Ù│┤Û│áÙÉ£ empagliflozinýùÉ ÙîÇÝò£ ýòêýáòýä▒ ýù░ÛÁ¼ýØ© Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes trial (EMPA-REG OUTCOME) Û▓░Û│╝ÙÑ╝ Ù│┤Ù®┤ empagliflozinýØä Ýê¼ýò¢Ýò£ ÛÁ░ýùÉýä£ ÙîÇýí░ÛÁ░ýùÉ Ù╣äÝò┤ 3-point MACEÛ░Ç 14% Û░ÉýåîÙÉÿýùêÛ│á ýï¼ÝÿêÛ┤ÇÛ│ä ýé¼ÙºØ Ùï¿ÙÅàýùÉ ÙîÇÝò┤ýä£Ùèö 38%ýØÿ Û░ÉýåîÙÑ╝ Ù│┤ýÿÇÙïñ[20]. ÙÿÉÝò£, CanagliflozinýØä ýé¼ýÜ®Ýò£ Canagliflozin Cardiovascular Assessment Study (CANVAS) ýù░ÛÁ¼ýùÉýä£ÙÅä Ùºêý░¼Û░ÇýºÇÙí£ 3-point MACEýùÉ ÙîÇÝò┤ ý£áýØÿÝò£ Û░ÉýåîÙÑ╝ Ù│┤ýÿÇý£╝Ù®░ Ùºîýä▒ ýïáÙÂÇýáä ÝÖÿý×ÉÙÑ╝ ÙîÇýâüý£╝Ùí£ Ýò£ CANVASRenal (CANVAS-R) Ù░Å Canagliflozin and Renal Events in Diabetes with Established Nephropathy Clinical Evaluation (CREDENCE) ýù░ÛÁ¼ýùÉýä£Ùèö canagliflozin Ýê¼ýò¢ÛÁ░ýùÉýä£ ÙîÇýí░ÛÁ░ýùÉ Ù╣äÝòÿýù¼ ý£áýØÿÝòÿÛ▓î ý£äÝùÿýä▒ýØ┤ Û░ÉýåîÝòÿýÿÇÙïñ[21-23]. Ýò£ÝÄ©, dapagliflozin Ýê¼ýò¢ÛÁ░Û│╝ ÙîÇýí░ÛÁ░ýØä Ù╣äÛÁÉÝò£ ýù░ÛÁ¼(dapagliflozin effect on cardiovascular events-thrombosis in myocardial infarction 58, DECLARE-TIMI 58)ýùÉýä£Ùèö 3-point MACEýØÿ Ù░£ýâØýØä ý£áýØÿÝòÿÛ▓î Û░Éýåîýï£ÝéñýºÇÙèö Ù¬╗ÝòÿýÿÇý£╝Ùéÿ ýï¼ÝÿêÛ┤ÇÛ│ä ýé¼ÙºØÙÑáÛ│╝ ýï¼ÙÂÇýáäýùÉ ýØÿÝò£ ý×àýøÉ Û░üÛ░üýùÉ ÙîÇÝò┤ýä£Ùèö ý£áýØÿÝò£ Û░ÉýåîÙÑ╝ ÝÖòýØ©ÝòÿýÿÇÙïñ[24]. ýØ┤ 3Û░ÇýºÇ ýò¢ýá£ýùÉ ÙîÇÝò£ ý×äýâü ýù░ÛÁ¼ýØÿ Û▓░Û│╝Ùí£ SGLT-2 ýûÁýá£ýá£Ùèö ýï¼ÝÿêÛ┤Ç ýòêýáòýä▒ýØÇ Ù¼╝ÙíáýØ┤Û│á ýïáý×Ñ ýºêÝÖÿý×ÉýùÉ ÙîÇÝò£ ýòêýáòýä▒ÙÅä Ù│┤ýù¼ýú╝ýùêý£╝Ù®░, ýï¼ÝÿêÛ┤Ç ýºêÝÖÿ ýÿêÙ░®ÝÜ¿Û│╝ ýù¡ýï£ Ùø░ýû┤Ùé¿ýØä Ù│┤ýù¼ýú╝ýùêÙïñ(Fig. 1). Ýè╣Ý×ê, SGLT-2 ýûÁýá£ýá£Ùèö ÝÿêÙï╣ýØÿ Û░Éýåî ýÖ©ýùÉÙÅä ýé╝Ýê¼ýòòýä▒ýØ┤Ùç¿(osmotic diuresis) Ù░Å ÙéÿÝè©ÙÑ¿ýä▒ýØ┤Ùç¿(natriuresis)Ùí£ ýØ©Ýò£ ýñæÙô▒ÙÅäýØÿ ýØ┤Ùç¿ ÝÜ¿Û│╝ÙÑ╝ Ù│┤ýù¼ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿÛ│╝ Ù│äÛ░£Ùí£ ýï¼ÙÂÇýáä ÝÖÿý×ÉýùÉýä£ Û©ìýáòýáü ý×æýÜ®ýØä ÝòÿÙª¼ÙØ╝ Û©░ÙîÇÝòÿýÿÇÙìÿ ýò¢ýá£Ùí£ ýïñýᣠýòêýáòýä▒ ýù░ÛÁ¼ýùÉýä£ ýï¼ÙÂÇýáäýùÉ ÙîÇÝò£ Û©ìýáòýáü ÝÜ¿Û│╝Û░Ç ÝÖòýØ©ÙÉÿÙ®┤ýä£ SGLT-2 ýûÁýá£ýá£ýØÿ ýï¼ÙÂÇýáäýùÉ ÙîÇÝò£ ÝÜ¿Û│╝ÙÑ╝ ÝÖòýØ©ÝòÿÙèö ý×äýâü ýù░ÛÁ¼Û░Ç ýºäÝûëÙÉÿýùêÙïñ. DapagliflozinýØÿ ýï¼ÙÂÇýáäýùÉ ÙîÇÝò£ ÝÜ¿Û│╝ÙÑ╝ ÝÖòýØ©ÝòÿýÿÇÙìÿ Dapagliflozin in Patients with Heart Failure and Reduced Ejection Fraction (DAPA-HF) ýù░ÛÁ¼ýùÉýä£ ýóîýï¼ýïñ Ù░òý£ÙÑáýØ┤ Û░ÉýåîÙÉ£ ýï¼ÙÂÇýáä ÝÖÿý×ÉýùÉýä£ dapagliflozinýØ┤ ýï¼ÝÿêÛ┤ÇÛ│ä ýé¼ÙºØ Ù░Å ýï¼ÙÂÇýáäýùÉ ýØÿÝò£ ý×àýøÉýØä Û░Éýåîýï£ÝéñÙèö Û▓âýØä ÝÖòýØ©ÝòÿýÿÇÛ│á ÙîÇýâü ÝÖÿý×ÉýØÿ 50%Û░Ç Ù╣ä Ùï╣Ùç¿ ÝÖÿý×ÉÙí£ Ùï╣Ùç¿ýØÿ ý£áÙ¼┤ ýâüÛ┤ÇýùåýØ┤ ÛÀ© ÝÜ¿Û│╝Û░Ç ý×êýØîýØ┤ ý×àýªØÙÉÿýùêÙïñ. ýØ┤Ýøä ýºäÝûëÙÉ£ empagliflozinýØÿ ýï¼ÙÂÇýáäýùÉ ÙîÇÝò£ ÝÜ¿Û│╝ÙÑ╝ Ù│© Cardiovascular and Renal Outcomes with Empagliflozin in Heart Failure (EMPEROR-REDUCED) ýù░ÛÁ¼ýùÉýä£ÙÅä Ùºêý░¼Û░ÇýºÇýØÿ Û▓░Û│╝ÙÑ╝ ý×àýªØÝò¿ý£╝Ùí£ýì¿ SGLT-2 ýûÁýá£ýá£Ùèö Ùï╣Ùç¿ ý╣ÿÙúîýò¢ýá£ÙÑ╝ Ùäÿýû┤ ýâêÙí£ýÜ┤ ýï¼ÙÂÇýáä ý╣ÿÙúîýò¢ýá£Ùí£ Û░üÛ┤æÙ░øÛ▓î ÙÉÿýùêÙïñ(Table 3) [25,26]. ÙÿÉÝò£, SGLT-2 ýûÁýá£ýá£ÙÅä GLP-1 ýêÿýÜ®ý▓┤ ý×æýÜ®ýá£ý▓ÿÙƒ╝ ýØ©ýèÉÙª░ ýáÇÝò¡ýä▒ Û░£ýäá, ý▓┤ýñæÛ░Éýåî Ùô▒ýØÿ ÝÜ¿Û│╝Û░Ç ý×êýû┤ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿ ý£äÝùÿýØ©ý×ÉýØÿ Û░£ýäáýùÉ ÙÅäýøÇýØä ýú╝Ù®░ ýØ┤Ùí£ ýØ©Ýò£ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿýØÿ ýÿêÙ░® ÝÜ¿Û│╝ÙÅä Ù│┤ýØ©Ùïñ[19]. ýØ┤Ùƒ¼Ýò£ Û▓░Û│╝Ùí£ ýØ©Ýò┤ ýòäýºü Û░ÇýØ┤Ùô£ÙØ╝ýØ©ýùÉ Ù░ÿýÿüÙÉÿýºÇÙèö ýòèýòÿý£╝Ùéÿ ÛÁ¡Ùé┤ýÖ© ýáäÙ¼©Û░ÇÙôñýØÇ Û©░ýáÇ ýï¼ý×Ñ ýºêÝÖÿýØ┤ ý×êÙèö ÝÖÿý×ÉýùÉýä£ SGLT-2 ýûÁýá£ýá£ÙÑ╝ Ù®öÝè©ÝżÙÑ┤Ù»╝ ÙîÇýïá Ùï╣Ùç¿ ý╣ÿÙúîýØÿ ýØ╝ý░¿ ýò¢ýá£Ùí£ Û│áÙáñÝòá Û▓âýØä ÛÂîÛ│áÝòÿÛ│á ý×êÙïñ[25,26].
ýØ┤Ùƒ¼Ýò£ ýù░ÛÁ¼ Û▓░Û│╝ÙÑ╝ Ù░öÝâòý£╝Ùí£ ÙîÇÝò£Ùï╣Ùç¿Ù│æÝòÖÝÜîýùÉýä£Ùèö Û┤ÇýâüÙÅÖÙºÑ ýºêÝÖÿ, ýï¼ÛÀ╝Û▓¢ýâë Ùô▒Û│╝ Û░ÖýØÇ ýï¼ý×Ñ ýºêÝÖÿýØ┤ ý×êÙèö Û│áý£äÝùÿÛÁ░ýùÉýä£Ùèö SGLT-2 ýûÁýá£ýᣠýñæ ýï¼ÝÿêÛ┤Ç ýÿêÙ░®ÝÜ¿Û│╝Û░Ç ý×àýªØÙÉ£ ýò¢ýá£ÙÑ╝ ýÜ░ýäá Û│áÙáñÝòÿÙÅäÙíØ ÛÂîÛ│áÝòÿÛ│á ý×êÛ│á Ù»©ÛÁ¡Û│╝ ý£áÙƒ¢Ùï╣Ùç¿Ù│æÝòÖÝÜîýùÉýä£ÙÅä ýú¢ýâüÛ▓¢ÝÖöýä▒ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿ(atherosclerotic cardiovascular disease, ASCVD)ýØä ÙÅÖÙ░ÿÝò£ Ùï╣Ùç¿Ù│æ ÝÖÿý×ÉýùÉÛ▓îÙèö SGLT-2 ýûÁýá£ýᣠÝÿ╣ýØÇ GLP-1 ýêÿýÜ®ý▓┤ ý×æýÜ®ýá£ÙÑ╝ ý▓ÿÙ░®ÝòÿÙèö Û▓âýØä ÛÂîÛ│áÝòÿÛ│á ý×êÙïñ[1]. Û▓îÙïñÛ░Ç Ù»©ÛÁ¡ FDAýùÉýä£Ùèö dapagliflozinýØä Ùï╣Ùç¿Ù│æ ÝÖÿý×ÉýØÿ ýï¼ÙÂÇýáä ýÿêÙ░®ýò¢ Ù░Å ýï¼ÙÂÇýáä ý╣ÿÙúîýá£Ùí£, canagliflozinýØä Ùï╣Ùç¿Ù│æýä▒ ýïáýºêÝÖÿýØä ÙÅÖÙ░ÿÝò£ ýá£2Ýÿò Ùï╣Ùç¿Ù│æ ý╣ÿÙúîýá£Ùí£ ýè╣ýØ©Ýò£ Ù░ö ý×êÙïñ. ÙïñÙºî, ýòäýºüÛ╣îýºÇ ÛÁ¡Ùé┤ Ù│┤Ýùÿ Û©ëýù¼ Û©░ýñÇýØÇ Ùï╣Ùç¿ ÝÖÿý×ÉýùÉýä£ Ù®öÝè©ÝżÙÑ┤Ù»╝ Ýÿ╣ýØÇ ýØ©ýèÉÙª░Û│╝ Ù│æýÜ® Ýê¼ýò¢ÝòÿÛ▒░Ùéÿ Ùï╣ÝÖöÝÿêýâëýåî(HbA1c)Û░Ç 7.5% ýØ┤ýâüýØ© Û▓¢ýÜ░Ùºî Û©ëýù¼ ýØ©ýáòýØ┤ ÙÉÿÛ│á, Ùï╣ÝÖöÝÿêýâëýåî(HbA1c)Û░Ç 7.5% Ù»©ÙºîýØÿ Û▓¢ýÜ░ ÝÖÿý×É Ù│©ýØ© ýáäýòíÙÂÇÙï┤ý£╝Ùí£ ý▓ÿÙ░®ýØ┤ Û░ÇÙèÑÝò£ ýâüÝâ£ýØ┤Ùïñ. Ùö░ÙØ╝ýä£ ýòäýºüÛ╣îýºÇÙèö ýØ┤ÙÑ╝ ý×äýâüýùÉ ýáüýÜ®ÝòÿÛ©░ýùÉÙèö ýá£Ýò£ýáÉýØ┤ ÙºÄÙïñ.
Ùï╣Ùç¿ ý╣ÿÙúîýò¢ýá£ýØÿ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿ ýòêýáòýä▒ ýù░ÛÁ¼Ùèö ý┤êÛ©░ýùÉÙèö ý×áý×¼ýáüý£╝Ùí£ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿýØÿ ý£äÝùÿýä▒ýØ┤ ý×êÙèö Ùï╣Ùç¿ ý╣ÿÙúîýò¢ýá£ÙÑ╝ ÙºëÛ©░ ý£äÝò┤ ýï£ý×æÙÉÿýùêý£╝Ùéÿ Û▓░ÛÁ¡ýùÉÙèö ýâêÙí£ýÜ┤ ýò¢ýá£ÙôñýØÿ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿ ýÿêÙ░®ÝÜ¿Û│╝Û░Ç ý×àýªØÙÉÿÙ®┤ýä£ Ùï╣Ùç¿Ù│æÙ┐ÉÙºî ýòäÙïêÙØ╝ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿ ý╣ÿÙúî ýºÇý╣¿ýùÉÙÅä Ù│ÇÝÖöÙÑ╝ ÙÂêÙƒ¼ýÿñÛ│á ý×êÙïñ. Ýÿäý×¼Û╣îýºÇýØÿ ýù░ÛÁ¼ Û▓░Û│╝Ùí£ Ù│┤ýòä DPP-4 ýûÁýá£ýá£Ùèö ýï¼ÝÿêÛ┤Ç ýòêýáòýä▒ýØÇ ýªØÙ¬àÙÉÿýùêý£╝Ùéÿ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿýùÉ ÙîÇÝò£ ýÿêÙ░®ÝÜ¿Û│╝Ùèö ÝÖòýïñý╣ÿ ýòèý£╝Ù®░, ýØ╝ÙÂÇ ýò¢ýá£ýØÿ Û▓¢ýÜ░ ýï¼ÙÂÇýáä ýòàÝÖöýØÿ ý£äÝùÿýä▒ýØ┤ ý×êÙïñ. Ù░ÿÙ®┤, 3Û░£ýØÿ SGLT-2 ýûÁýá£ýá£(empagliflozin, canagliflozin, dapagliflozin)ýÖÇ 4Û░£ýØÿ GLP-1 ýêÿýÜ®ý▓┤ ý×æýÜ®ýá£(liraglutide, albiglutide, semaglutide, dulaglutide)Ùèö ýï¼ÝÿêÛ┤Ç ýòêýáòýä▒ýØ┤ ÝÖòýØ©ÙÉÿýùêýØä Ù┐É ýòäÙïêÙØ╝ ý£áýØÿÝòÿÛ▓î ýï¼ÝÿêÛ┤Ç ýºêÝÖÿýØÿ Ù░£ýâØýØä Û░Éýåîýï£Ýé¿ÙïñÙèö Û▓âýØ┤ ÝÖòýØ©ÙÉÿýû┤ ýØ┤ ÙæÉ Û░ÇýºÇ ýò¢ýá£ýØÿ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿ ýÿêÙ░®ÝÜ¿Û│╝Ùèö ÙÂäÙ¬àÝòÿÙïñ[27]. Ýè╣Ý×ê SGLT-2 ýûÁýá£ýá£Ùèö Ùºîýä▒ ýïáÙÂÇýáä ÝÖÿý×ÉýùÉýä£ ýïáÛ©░ÙèÑ Û░£ýäáýØÿ ÝÜ¿Û│╝ÙÑ╝ Ù│┤ýØ┤Û│á Ù╣ä Ùï╣Ùç¿ ÝÖÿý×ÉýùÉýä£ÙÅä ýï¼ÙÂÇýáäýùÉ ýØÿÝò£ ý×àýøÉÙÑá Û░Éýåî Ù░Å ýïáý×ÑÛ©░ÙèÑ Û░£ýäáýùÉÙÅä ÝÜ¿Û│╝Û░Ç ý×êý£╝Ù®░ Ýè╣Ý×ê ýóîýï¼ýïñ Ù░òý£ýØ┤ Û░ÉýåîÙÉ£ ýï¼ÙÂÇýáä ÝÖÿý×ÉýùÉýä£ ýï¼ÝÿêÛ┤ÇÛ│ä ýé¼ÙºØ Ù░Å ýï¼ÙÂÇýáäýùÉ ýØÿÝò£ ý×àýøÉýØä Û░Éýåîýï£ÝéñÙèö Û▓░Û│╝ÙÑ╝ Ù│┤ýù¼ ý×äýâüýáüý£╝Ùí£ ýù¼Ùƒ¼ ÝÖÿý×ÉÛÁ░ýùÉýä£ ýä£Ùí£ ÙïñÙÑ© ÝÜ¿Û│╝ÙÑ╝ Û©░ÙîÇÝòÿÙ®░ ýáüýÜ®Ýòá ýêÿ ý×êÛ▓áÙïñ. Ùö░ÙØ╝ýä£ Ýè╣ýáò ÝÖÿý×ÉÛÁ░ýùÉýä£ SGLT-2 ýûÁýá£ýá£ýØÿ ÝÜ¿Û│╝ÙÑ╝ ÝÖòýØ©ÝòÿÙèö ýù░ÛÁ¼ÙôñýØ┤ ÝòäýÜöÝòá Û▓âý£╝Ùí£ ýâØÛ░üÙÉÿÙ®░, Ýÿäý×¼ ýóîýï¼ýïñ Ù░òý£ÙÑáýØ┤ Ù│┤ýí┤ÙÉ£ ýï¼ÙÂÇýáä ÝÖÿý×ÉÙéÿ Û©ëýä▒ ýï¼ÛÀ╝Û▓¢ýâë ÝÖÿý×É, Û┤ÇýâüÙÅÖÙºÑ ýºêÝÖÿ ÝÖÿý×ÉÙÑ╝ ÙîÇýâüý£╝Ùí£ Ýò£ ýù░ÛÁ¼Û░Ç ýºäÝûëÙÉÿýû┤ Û│º Ù░£Ýæ£ ýÿêýáòýùÉ ý×êÙïñ. ÙÿÉÝò£, GLP-1 ýêÿýÜ®ý▓┤ ý×æýÜ®ýá£Ùèö ý▓┤ýñæÛ░ÉÙƒëýùÉ ý×êýû┤ Ùø░ýû┤Ùé£ ÝÜ¿Û│╝ÙÑ╝ Ù│┤ýù¼ ýâêÙí£ýÜ┤ Ù╣äÙºî ý╣ÿÙúîýá£Ùí£ ÙûáýÿñÙÑ┤Û│á ý×êýû┤ ÝûÑÝøä Ùï╣Ùç¿ ý╣ÿÙúîýò¢ýá£ÙÑ╝ Ùäÿýû┤ýäá ýâêÙí£ýÜ┤ ýÿüýù¡ýØÿ ý╣ÿÙúîýá£Ùí£ýä£ýØÿ ýù¡ÝòáÙÅä Û©░ÙîÇÙÉ£Ùïñ. Ýò£ÝÄ©, SGLT-2 ýûÁýá£ýá£ýÖÇ GLP-1 ýêÿýÜ®ý▓┤ ý×æýÜ®ýá£ÙÑ╝ Ýò¿Û╗ÿ Ýê¼ýò¢ÝòÿýÿÇýØä ÙòîýØÿ ýÂöÛ░ÇýáüýØ© ÝÜ¿Û│╝Ùèö ýòäýºü ýòîÙáñýºä Ù░öÛ░Ç ýùåýû┤ ÝûÑÝøä ýØ┤ýùÉ ÙîÇÝò£ ýù░ÛÁ¼ÙÅä ÝòäýÜöÝòá Û▓âý£╝Ùí£ Ù│┤ýØ©Ùïñ.
Ùï╣Ùç¿ ý╣ÿÙúîýò¢ýá£ýØÿ ýï¼ÝÿêÛ┤ÇÛ│ä ýºêÝÖÿ ýòêýáòýä▒ ýù░ÛÁ¼Ùèö ý┤êÛ©░ýùÉ ý×áý×¼ýáüý£╝Ùí£ ý£áÝò┤Ýò£ ýò¢ýá£ÙÑ╝ ÝÖòýØ©ÝòÿÙèö Û▓âýùÉýä£ ÝÖòý×ÑÙÉÿýû┤ ýâêÙí£ýÜ┤ ýò¢ýá£ýùÉ ÙîÇÝò£ ýù░ÛÁ¼ Û▓░Û│╝Ùí£ ýØ©Ýò┤ Ùï╣Ùç¿ Ù░Å ýï¼ÝÿêÛ┤Ç ÝÖÿý×ÉýùÉýä£ ý×àýªØÙÉ£ ýòêýáòýä▒Ù┐ÉÙºî ýòäÙïêÙØ╝ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿýØä ýÿêÙ░® Ýÿ╣ýØÇ ý╣ÿÙúîÝòÿÙèö ÙÂäýò╝Û╣îýºÇ ÝÖòý×ÑÙÉÿýùêÙïñ. ýØ┤ýÖÇ Û░ÖýØÇ Û▓░Û│╝Ùèö Ùï╣Ùç¿ ý╣ÿÙúîýò¢ýá£ýØÿ ýäáÝâØýùÉ ý×êýû┤ ýï¼ÝÿêÛ┤Ç ýòêýáòýä▒ Ù░Å ýï¼ÝÿêÛ┤Ç ý£áÝÜ¿ýä▒ýØÿ ýªØÛ▒░Û░Ç ý×êÙèö ýò¢ýᣠÝè╣Ý×ê GLP-1 ýêÿýÜ®ý▓┤ ý×æýÜ®ýá£ýÖÇ SGLT-2 ýûÁýá£ýá£ÙÑ╝ ýÂöý▓£Ýò┤ýò╝ ÝòÿÙèö ý╣ÿÙúî Ýî¿Ùƒ¼Ùïñý×äýØÿ Ù│ÇÝÖöÙÑ╝ Û░Çýá©ýÖöÙïñ. ÝûÑÝøä ýÂöÛ░ÇýáüýØ© ýù░ÛÁ¼ÙÑ╝ ÝåÁÝò┤ ÝÖÿý×ÉýØÿ ýï¼ÝÿêÛ┤Ç ýºêÝÖÿýØÿ ý£äÝùÿÙÅä, ýºêÝÖÿ ý£áÙ¼┤ýùÉ Ùö░ÙÑ© ýò¢ýᣠýäáÝâØýØ┤ Û░ÇÙèÑÝò┤ýºê Û▓âý£╝Ùí£ ýÿêýâüÙÉÿÙ®░ Ùï╣Ùç¿Ù│æ ý╣ÿÙúî Ù¬®Ýæ£Û░Ç Ùï¿ýê£Ýò£ Ùï╣Ùç¿ ýí░ýáêýØ┤ ýòäÙïî Ùìö ÙéÿýòäÛ░Ç ÝÜ¿Û│╝ýáüýØ© ýï¼ÝÿêÛ┤Ç ýºêÝÖÿýØÿ ýÿêÙ░® Ù░Å ýé¼ÙºØÙÑá Û░ÉýåîýùÉ ýØ┤ÙÑ┤Û▓î ÙÉá Û▓âý£╝Ùí£ ýÿêýâüÙÉ£Ùïñ.
REFERENCES
1. American Diabetes Association. 10. Cardiovascular disease and risk management: standards of medical care in diabetes-2021. Diabetes Care 2021;44 Suppl 1:S125ÔÇôS150.

2. Food and Drug Administration. Guidance for industry: diabetes mellitusÔÇòevaluating cardiovascular risk in new antidiabetic therapies to treat type 2 diabetes [Internet]. Silver Spring (MD): Food and Drug Administration, Center for Drug Evaluation and Research (CDER). c2008 [cited 2021 Feb 24]. Available from: https://www.fda.gov/media/71297/download
3. Lincoff AM, Wolski K, Nicholls SJ, Nissen SE. Pioglitazone and risk of cardiovascular events in patients with type 2 diabetes mellitus: a meta-analysis of randomized trials. JAMA 2007;298:1180ÔÇô1188.


4. Bonaventura A, Carbone S, Dixon DL, Abbate A, Montecucco F. Pharmacologic strategies to reduce cardiovascular disease in type 2 diabetes mellitus: focus on SGLT-2 inhibitors and GLP-1 receptor agonists. J Intern Med 2019;286:16ÔÇô31.


5. Dicker D. DPP-4 inhibitors: impact on glycemic control and cardiovascular risk factors. Diabetes Care 2011;34 Suppl 2:S276ÔÇôS278.


6. Rosenstock J, Perkovic V, Johansen OE, et al. Effect of linagliptin vs placebo on major cardiovascular events in adults with type 2 diabetes and high cardiovascular and renal risk: the CARMELINA randomized clinical trial. JAMA 2019;321:69ÔÇô79.


7. Scirica BM, Bhatt DL, Braunwald E, et al. Saxagliptin and cardiovascular outcomes in patients with type 2 diabetes mellitus. N Engl J Med 2013;369:1317ÔÇô1326.


8. White WB, Cannon CP, Heller SR, et al. Alogliptin after acute coronary syndrome in patients with type 2 diabetes. N Engl J Med 2013;369:1327ÔÇô1335.


9. Green JB, Bethel MA, Armstrong PW, et al. Effect of sitagliptin on cardiovascular outcomes in type 2 diabetes. N Engl J Med 2015;373:232ÔÇô242.

10. Zannad F, Cannon CP, Cushman WC, et al. Heart failure and mortality outcomes in patients with type 2 diabetes taking alogliptin versus placebo in EXAMINE: a multicentre, randomised, double-blind trial. Lancet 2015;385:2067ÔÇô2076.


11. Doyle ME, Egan JM. Mechanisms of action of glucagon-like peptide 1 in the pancreas. Pharmacol Ther 2007;113:546ÔÇô593.


12. Marso SP, Daniels GH, Brown-Frandsen K, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2016;375:311ÔÇô322.



13. Marso SP, Bain SC, Consoli A, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med 2016;375:1834ÔÇô1844.


14. Hernandez AF, Green JB, Janmohamed S, et al. Albiglutide and cardiovascular outcomes in patients with type 2 diabetes and cardiovascular disease (harmony outcomes): a double-blind, randomised placebo-controlled trial. Lancet 2018;392:1519ÔÇô1529.

15. Gerstein HC, Colhoun HM, Dagenais GR, et al. Design and baseline characteristics of participants in the Researching cardiovascular Events with a Weekly INcretin in Diabetes (REWIND) trial on the cardiovascular effects of dulaglutide. Diabetes Obes Metab 2018;20:42ÔÇô49.


16. Gupta A, Jelinek HF, Al-Aubaidy H. Glucagon like peptide-1 and its receptor agonists: their roles in management of Type 2 diabetes mellitus. Diabetes Metab Syndr 2017;11:225ÔÇô230.


17. Pfeffer MA, Claggett B, Diaz R, et al. Lixisenatide in patients with type 2 diabetes and acute coronary syndrome. N Engl J Med 2015;373:2247ÔÇô2257.


18. Holman RR, Bethel MA, Mentz RJ, et al. Effects of once-weekly exenatide on cardiovascular outcomes in type 2 diabetes. N Engl J Med 2017;377:1228ÔÇô1239.


19. Abdul-Ghani MA, Norton L, Defronzo RA. Role of sodium-glucose cotransporter 2 (SGLT 2) inhibitors in the treatment of type 2 diabetes. Endocr Rev 2011;32:515ÔÇô531.


20. Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med 2015;373:2117ÔÇô2128.


21. Neal B, Perkovic V, Mahaffey KW, et al. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med 2017;377:644ÔÇô657.


22. Perkovic V, Jardine MJ, Neal B, et al. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. N Engl J Med 2019;380:2295ÔÇô2306.


23. Neal B, Perkovic V, Matthews DR. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med 2017;377:2099.

24. Wiviott SD, Raz I, Bonaca MP, et al. Dapagliflozin and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2019;380:347ÔÇô357.


25. McMurray JJV, Solomon SD, Inzucchi SE, et al. Dapagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med 2019;381:1995ÔÇô2008.

Cardiovascular outcome trials of SGLT2-Inhibitors. EMPA-REG OUTCOME trial for empagliflozin, CANVAS trial for canagliflozin, DECLARE-TIMI 58 trial for dapagliflozin. All relative risk reduction is statistically significant (p < 0.05) except CV death in DECLARE-TIMI 58. 3-point MACE, composite of death from cardiovascular causes, nonfatal myocardial infarction, or nonfatal stroke; CV, cardiovascular; HF, heart failure.
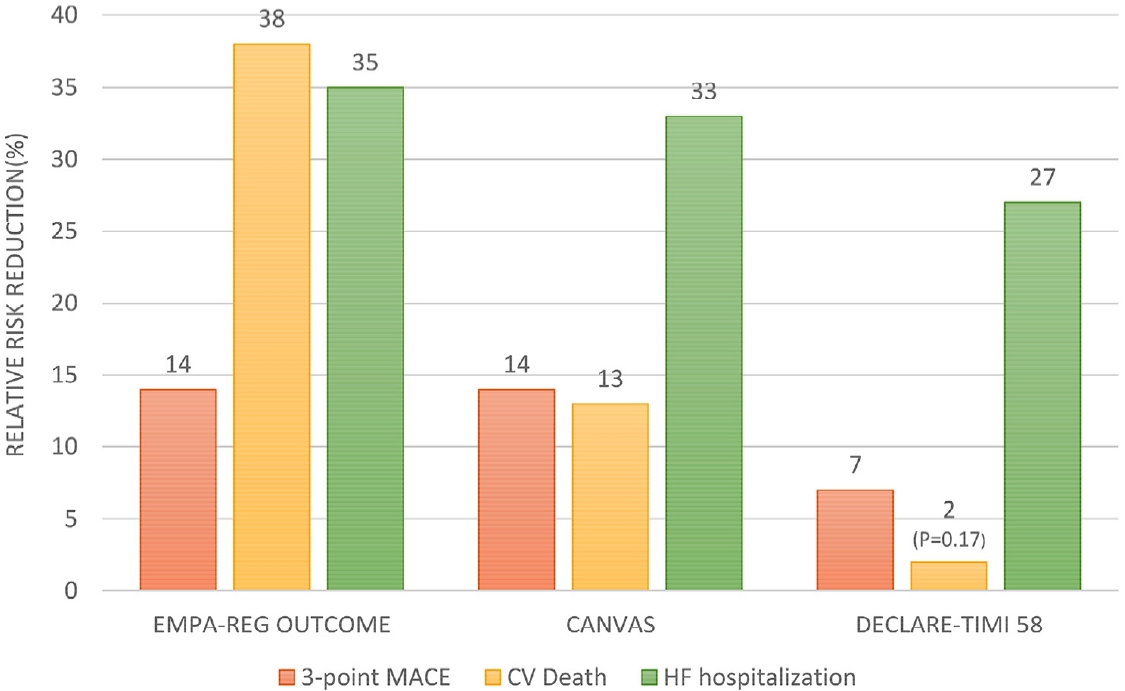
Figure 1.
Table 1.
Cardiovascular outcome trials of DPP-4 inhibitors
| Saxagliptin (SAVOR-TIMI 53) | Alogliptin (EXAMINE) | Sitagliptin (TECOS) | Linagliptin (CARMELINA) | |
|---|---|---|---|---|
| Main inclusion criteria | Type 2 diabetes, History of or multiple risk factors for CVD | Type 2 diabetes, ACS within 15ÔÇô90 days before randomization | Type 2 diabetes, Preexisting CVD | Type 2 diabetes, High CV and renal risk |
| Primary outcome (3-point MACE)a | HR 1.00 (0.89ÔÇô1.12) | HR 0.96 (95% UL Ôëñ1.16) | HR 0.98 (0.89ÔÇô1.08) | HR 1.02 (0.89ÔÇô1.17) |
| CV death | HR 1.03 (0.87ÔÇô1.22) | HR 0.85 (0.66ÔÇô1.10) | HR 1.03 (0.89ÔÇô1.19) | HR 0.96 (0.81ÔÇô1.14) |
| Non-fatal MI | HR 0.95 (0.80ÔÇô1.12) | HR 1.08 (0.88ÔÇô1.33) | HR 0.95 (0.81ÔÇô1.11) | HR 1.12 (0.90ÔÇô1.40) |
| Non-fatal stroke | HR 1.11 (0.88ÔÇô1.39) | HR 0.91 (0.55ÔÇô1.50) | HR 0.97 (0.79ÔÇô1.19) | HR 0.91 (0.67ÔÇô1.23) |
| HF hospitalization | HR 1.27 (1.07ÔÇô1.51) | HR 1.07 (0.79ÔÇô1.46) | HR 1.00 (0.83ÔÇô1.20) | HR 0.90 (0.74ÔÇô1.08) |
| Unstable angina | HR 1.19 (0.89ÔÇô1.60) | HR 0.90 (0.60ÔÇô1.37) | HR 0.90 (0.70ÔÇô1.16) | HR 0.87 (0.57ÔÇô1.31) |
| All-cause mortality | HR 1.11 (0.96ÔÇô1.27) | HR 0.80 (0.62ÔÇô1.03) | HR 1.01 (0.90ÔÇô1.14) | HR 0.98 (0.84ÔÇô1.13) |
Table 2.
Cardiovascular outcome trials of GLP-1 agonists
| Liraglutide (LEADER) | Albiglutide (Harmony Outcomes) | Semaglutide (SUSTAIN-6) | Dulaglutide (REWIND) | Lixisenatide (ELIXA) | Exenatide (EXSCEL) | |
|---|---|---|---|---|---|---|
| Main inclusion criteria | Type 2 diabetes, Preexisting CVD, CKD, or HF at  50 years of age or CV risk at  60 years of age | Type 2 diabetes, Preexisting CVD | Type 2 diabetes, Preexisting CVD, HF, or CKD at  50 years of age or CV risk at  60 years of age | Type 2 diabetes, Prior ASCVD event or risk factors for ASCVD | Type 2 diabetes, History of ACS (< 180 days) | Type 2 diabetes with or without preexisting CVD |
| Primary out- come (3-point MACE)a | HR 0.87 (0.78ÔÇô0.97) | HR 0.78 (0.68ÔÇô0.90) | HR 0.74 (0.58ÔÇô0.95) | HR 0.88 (0.79ÔÇô0.99) | HR 1.02 (0.89ÔÇô1.17) | HR 0.91 (0.83ÔÇô1.00) |
| CV death | HR 0.78 (0.66ÔÇô0.93) | HR 0.93 (0.73ÔÇô1.19) | HR 0.98 (0.65ÔÇô1.48) | HR 0.91 (0.78ÔÇô1.06) | HR 0.98 (0.78ÔÇô1.22) | HR 0.88 (0.76ÔÇô1.02) |
| Non-fatal MI | HR 0.86 (0.73ÔÇô1.00) | HR 0.75 (0.61ÔÇô0.90) | HR 0.74 (0.51ÔÇô1.08) | HR 0.96 (0.79ÔÇô1.15) | HR 1.03 (0.87ÔÇô1.22) | HR 0.97 (0.85ÔÇô1.10) |
| Non-fatal stroke | HR 0.86 (0.71ÔÇô1.06) | HR 0.86 (0.66ÔÇô1.14) | HR 0.61 (0.38ÔÇô0.99) | HR 0.76 (0.61ÔÇô0.95) | HR 1.12 (0.79ÔÇô1.58) | HR 0.85 (0.70ÔÇô1.03) |
| HF hospitalization | HR 0.87 (0.73ÔÇô1.05) | - | HR 1.11 (0.77ÔÇô1.61) | HR 0.93 (0.77ÔÇô1.12) | HR 0.96 (0.78ÔÇô1.23) | HR 0.94 (0.78ÔÇô1.13) |
| Unstable angina | HR 0.98 (0.76ÔÇô1.26) | - | HR 0.82 (0.47ÔÇô1.44) | HR 1.14 (0.84ÔÇô1.54) | HR 1.11 (0.47ÔÇô2.62) | HR 1.05 (0.94ÔÇô1.18) |
| All-cause mortality | HR 0.85 (0.74ÔÇô0.97) | HR 0.95 (0.79ÔÇô1.16) | HR 1.05 (0.74ÔÇô1.50) | HR 0.90 (0.80ÔÇô1.01) | HR 0.94 (0.78ÔÇô1.13) | HR 0.86 (0.77ÔÇô0.97) |
Outcomes reported as adjusted hazard ratios (HRs) with 95% confidence interval.
-, not assessed/reported; CVD, cardiovascular disease; CKD, chronic kidney disease; HF, Heart failure; CV, cardiovascular; ASCVD, atherosclerotic cardiovascular disease; MI, myocardial infarction.
a Primary outcome was a composite of 3-point MACE (death from cardiovascular causes, nonfatal myocardial infarction, or nonfatal stroke) in all trials except Harmony Outcomes and ELIXA, in which primary outcome was a composite of 4-point MACE (death from cardiovascular causes, nonfatal myocardial infarction, nonfatal stroke, or unstable angina).
Table 3.
SGLT-2 inhibitors in patients with reduced ejection fraction: the results of DAPA-HF and EMPEROR-REDUCED Trial
-
METRICS

-
- 1 Crossref
- 0 Scopus
- 2,849 View
- 353 Download



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



