 |
 |
| Korean J Med > Volume 97(6); 2022 > Article |
|
Abstract
Chronic hepatitis B virus (HBV) infection is a major cause of liver cirrhosis and hepatocellular carcinoma in Korea. While the prevalence of HBV infection is gradually declining in Korea, 3 of the population still suffers from HBV-related chronic liver diseases. In this review, we summarize the updated clinical guidelines for management of chronic HBV, as revised by the Korean Association for the Study of the Liver in 2022.
Ùºîýä▒ BÝÿòÛ░äýù╝ýØÇ BÝÿòÛ░äýù╝ Ù░öýØ┤Ùƒ¼ýèñ(hepatitis B virus, HBV) Û░Éýù╝ýØ┤ 6Û░£ýøö ýØ┤ýâü ýºÇýåìÙÉÿÙ®┤ýä£ Û░äýùÉ Ùºîýä▒ ýù╝ýªØ Û┤┤ýé¼Û░Ç Ù░£ýâØÝòÿÙèö ýºêÝÖÿýØ┤Ùïñ. ÛÁ¡Û░ÇýÿêÙ░®ýáæýóà ýé¼ýùà Ù░Å ýú╝ýé░Û©░ Û░Éýù╝ ýÿêÙ░®ýé¼ýùàý£╝Ùí£ ýØ©Ýò┤ ýÜ░Ùª¼ÙéÿÙØ╝ýØÿ HBV Û░Éýù╝ý£¿ýØÇ ýáÉý░¿ Û░ÉýåîÝòÿýù¼ 3%ÙÑ╝ ý£áýºÇÝòÿÛ│á ý×êÙïñ. ÝòÿýºÇÙºî BÝÿòÛ░äýù╝ýØÇ Ýÿäý×¼ÙÅä ýÜ░Ùª¼ÙéÿÙØ╝ Ùºîýä▒ Û░äýºêÝÖÿýØÿ ýò¢ 70%ÙÑ╝ ý░¿ýºÇÝòÿÙèö ýú╝ýÜö ýøÉýØ©ýØ┤Ù®░, ÙåÆýØÇ Û░äýºêÝÖÿ Ù░Å Û░äýòö Û┤ÇÙá¿ ýé¼ÙºØÙÑáÙí£ ýØ©Ýò┤ ýù¼ýáäÝ×ê ÛÁ¡Ù»╝ Û▒┤Û░òýØÿ ý£äÝÿæ ýÜöýåîÙí£ Ùé¿ýòäý×êÙïñ[1].
ýÁ£ÛÀ╝ BÝÿòÛ░äýù╝ Ýò¡Ù░öýØ┤Ùƒ¼ýèñýá£ýØÿ ýºÇýåìýáüýØ© Ù░£ýáäÛ│╝ ýù¼Ùƒ¼ Û░ÇýºÇ ýù░ÛÁ¼ýùÉýä£ ýêÿýºæÙÉÿÙèö ýªØÛ▒░ÙÑ╝ Ù░öÝâòý£╝Ùí£ ÙîÇÝò£Û░äÝòÖÝÜî Ùºîýä▒ BÝÿòÛ░äýù╝ ýºäÙúîýºÇý╣¿ýØÇ Û¥©ýñÇÝ×ê Û░£ýáòÙÉÿÛ│á ý×êÙïñ. Ùºîýä▒ BÝÿòÛ░äýù╝ýùÉ Û┤ÇÝò£ ýáäÙ®┤ Û░£ýáòýØä Ùï¿ÝûëÝòÿýÿÇÙìÿ 2018Ùàä ýºäÙúîýºÇý╣¿Û│╝ Ùï¼Ùª¼, 2022Ùàä ýºäÙúîýºÇý╣¿ýØÇ ýÁ£ÛÀ╝ Ù░£Ýæ£ÙÉ£ ýØÿÝòÖýáòÙ│┤ÙÑ╝ Ù░öÝâòý£╝Ùí£ ýÁ£ýïá ýºÇÛ▓¼ýØÿ Ù░ÿýÿüýØ┤ ÝòäýÜöÝò£ ýú╝ýÜö 12Û░£ ý×äýâü ýú╝ýá£ÙÑ╝ ýäáÙ│äÝòÿÛ│á ýØ┤ýùÉ ÙîÇÝòÿýù¼ ýºäÙúî ÛÂîÛ│áýòêýØä ýêÿýáò Ù│┤ýÖäÝòÿÙèö ÙÂÇÙÂä Û░£ýáò Ù░®ýïØý£╝Ùí£ ýØ┤Ùú¿ýû┤ýíîÙïñ[2,3]. Ù│©Û│áýùÉýä£Ùèö 2022Ùàä ÙÂÇÙÂä Û░£ýáòÙÉ£ ÙîÇÝò£Û░äÝòÖÝÜî Ùºîýä▒ BÝÿòÛ░äýù╝ Û░ÇýØ┤Ùô£ÙØ╝ýØ©[3]ýØä ýñæýï¼ý£╝Ùí£, Ùºîýä▒ BÝÿòÛ░äýù╝ýØÿ ý×Éýù░Û▓¢Û│╝ýùÉýä£ ýñæýÜöýä▒ýØ┤ ÙîÇÙæÉÙÉÿÛ│á ý×êÙèö ýØ┤ÙÑ©Ù░ö ÔÇÿÝÜîýâëýºÇÙîÇ(grey zone)ÔÇÖ ÝÖÿý×ÉÙôñýùÉ ÙîÇÝò£ ýÁ£ÛÀ╝ ýù░ÛÁ¼ Û▓░Û│╝ Ù░Å ÛÂîÛ│áýé¼Ýò¡ÙôñýØä ýé┤ÝÄ┤Ù│┤Û│áý×É Ýò£Ùïñ. ÙÿÉÝò£, Ùé«ýØÇ ÙåìÙÅäýØÿ Ù░öýØ┤Ùƒ¼ýèñÝÿêýªØ ÝÖÿý×ÉýØÿ ý╣ÿÙúîýÖÇ Û▓¢ÛÁ¼ýÜ® Ýò¡Ù░öýØ┤Ùƒ¼ýèñýᣠý╣ÿÙúîÙÑ╝ ýóàÙúîÝòá ýêÿ ý×êÙèö Û©░ýñÇýùÉ ÙîÇÝò┤ ýåîÛ░£ÝòÿÛ│á Û░äýä©Ýżýòöýóà ÝÖÿý×É, ýïáÛ©░ÙèÑ ýØ┤ýâü ÙÿÉÙèö Û│¿ÙîÇýé¼ ýºêÝÖÿý×ÉýùÉýä£ýØÿ BÝÿòÛ░äýù╝ ý╣ÿÙúîýùÉ ÙîÇÝò┤ Û░äÙ×ÁÝ×ê ýòîýòäÙ│┤Û│áý×É Ýò£Ùïñ.
ýØ┤Ù▓ê 2022Ùàä ýºäÙúîýºÇý╣¿ýùÉýä£Ùèö Ùºîýä▒ BÝÿòÛ░äýù╝ýØÿ ý×Éýù░Û▓¢Û│╝ ýñæ ýû┤ÙèÉ Ýò£ Ùï¿Û│äýùÉ ýáòÝÖòÝ×ê ÙÂÇÝò®ÝòÿýºÇ ýòèÙèö ýâüÝâ£ýØ© ÔÇÿÝÜîýâëýºÇÙîÇÔÇÖýùÉ ÙîÇÝò┤ýä£ ý▓ÿýØîý£╝Ùí£ ýû©Û©ëÝòÿýÿÇÙïñ. Ùºîýä▒ BÝÿòÛ░äýù╝ýØÿ ý×Éýù░ Û▓¢Û│╝Ùèö Ù®┤ýù¡Û┤Çýܮ۩░, hepatitis B e antigen (HBeAg) ýûæýä▒ Ù®┤ýù¡ÝÖ£ÙÅÖÛ©░, Ù®┤ýù¡Ù╣äÝÖ£ÙÅÖÛ©░, HBeAg ýØîýä▒ Ù®┤ýù¡ÝÖ£ÙÅÖÛ©░, HBsAg ýåîýïñÛ©░Ùí£ ÙéÿÙêäýû┤ýºäÙïñ(Table 1). ýØ┤Ùƒ¼Ýò£ ý×Éýù░Û▓¢Û│╝ Ùï¿Û│äÙôñýØÇ ÝÖÿý×ÉÙºêÙïñ ýºÇýåì Û©░Û░äýØ┤ ÙïñýûæÝòÿÛ│á Ýò¡ýâü ýù░ýåìýáüýØ┤ýºÇ ýòèý£╝Ù®░, ýû┤ÙèÉ Ýò£ Ùï¿Û│äýùÉ ýáòÝÖòÝ×ê ÙÂÇÝò®ÝòÿýºÇ ýòèÙèö ÝÜîýâëýºÇÙîÇÛ░Ç ýí┤ý×¼Ýòá ýêÿ ý×êÙïñ. ýÿêÙÑ╝ ÙôñÙ®┤ HBeAg ýûæýä▒ ÝÖÿý×ÉýùÉýä£ Ýÿêý▓¡ HBV DNAÛ░Ç 107 IU/mL Ù»©ÙºîýØ┤Ù®┤ýä£ Ýÿêý▓¡ ALTÛ░Ç ýâüýè╣ÝòÿýºÇ ýòèÛ▒░Ùéÿ HBeAg ýØîýä▒ ÝÖÿý×ÉýùÉýä£ Ýÿêý▓¡ HBV DNAÛ░Ç 2,000 IU/mL ýØ┤ýâüýØ┤Ù®┤ýä£ Ýÿêý▓¡ ALTÛ░Ç ýâüýè╣ÝòÿýºÇ ýòèÙèö Û▓¢ýÜ░ýÖÇ Û░ÖýØ┤ ý×äýâüýáü ýºÇÝæ£ýØ© Ýÿêý▓¡ HBV DNAýÖÇ ALTÙÑ╝ Û©░ýñÇý£╝Ùí£ ýû┤ÙèÉ Ýò£ Ùï¿Û│äýùÉ ýáòÝÖòÝ×ê ÙÂÇÝò®ÝòÿýºÇ ýòèÙèö ýâüÝâ£ÙÑ╝ ÔÇÿÝÜîýâëýºÇÙîÇ(grey zone)ÔÇÖÙØ╝Û│á ýáòýØÿÝòá ýêÿ ý×êÙïñ. Ùºîýä▒ BÝÿòÛ░äýù╝ ÝÖÿý×ÉýØÿ ýò¢ 30%Û░Ç ÝÜîýâëýºÇÙîÇýùÉ Ýò┤Ùï╣ÝòÿÙèö Û▓âý£╝Ùí£ Ù│┤Û│áÙÉÿýû┤ ý×êý£╝Ù®░, ÝÜîýâëýºÇÙîÇ ÝÖÿý×ÉÙôñýØÿ Û▓¢ýÜ░ Û░äýåÉýâüýØä Ù░ÿýÿüÝòÿÙèö ýºÇÝæ£ýØ© Ýÿêý▓¡ ALTÛ░Ç ý£áýØÿÝòÿÛ▓î ÙåÆýºÇ ýòèýØÇ Û▓¢ýÜ░Û░Ç ÙºÄÙïñ[4,5]. ÝòÿýºÇÙºî ýâüÙîÇýáüý£╝Ùí£ ýù░Ùá╣ýØ┤ ÙåÆÛ│á, Ùï╣Ùç¿ Ù░Å ýºÇÙ░®Û░ä Ùô▒ýØÿ ÙÅÖÙ░ÿ ýºêÝÖÿ, BÝÿòÛ░äýù╝ Ù░öýØ┤Ùƒ¼ýèñ Ýè╣ýáò ÙÂÇý£äýØÿ Ù│ÇýØ┤ Ùô▒ Û░äýºêÝÖÿ ýºäÝûëÛ│╝ ýù░Û┤ÇÙÉ£ ýÂöÛ░ÇýáüýØ© ý£äÝùÿýØ©ý×ÉÙôñýØä ÙÅÖÙ░ÿÝòÿÙèö Û▓¢ýÜ░Û░Ç ý×êÙïñ.
ÝÜîýâëýºÇÙîÇýØÿ ÝÖÿý×ÉýùÉýä£ Ýò¡Ù░öýØ┤Ùƒ¼ýèñ ý╣ÿÙúî ýï£ý×æ ýù¼ÙÂÇÙÑ╝ Û▓░ýáòÝòÿÛ©░ ý£äÝò┤ Û░Çý×Ñ ýñæýÜöÝò£ Û▓âýØÇ Û░äýä¼ý£áÝÖöýÖÇ Û░äýØÿ ýù╝ýªØ ýáòÙÅäÙÑ╝ ýáòÝÖòÝòÿÛ▓î ÝÅëÛ░ÇÝòÿÙèö Û▓âýØ┤Ùïñ. Û░äýä¼ý£áÝÖöÙèö ý╣¿ýèÁýáüýØ© Û░äýâØÛ▓Ç ýÖ©ýùÉÙÅä, AST to Platelet Ratio Index (APRI), fibrosis-4 (FIB-4) index, Mac-2 binding protein glycosylation isomer (M2BPGi) Ùô▒ýØÿ Ýÿêý▓¡ Ýæ£ýºÇý×É[6,7] ÙÿÉÙèö Û░äýä¼ý£áÝÖöýèñý║ö(fibroscan), two-dimensional shear wave elastographyýÖÇ Û░ÖýØÇ Û░ä Ýâäýä▒ÙÅä Û▓Çýé¼Ùí£ Ù╣äý╣¿ýèÁýáüý£╝Ùí£ ÝÅëÛ░ÇÝòá ýêÿ ý×êÙïñ[8]. Û░äýâØÛ▓ÇýùÉýä£Ùèö F2 ýØ┤ýâüýØÿ ýä¼ý£áÝÖöÙÑ╝ ýØÿÙ»©ý×êÙèö ýä¼ý£áÝÖö(significant fibrosis)Ùí£ Û░äýú╝ÝòÿÙ®░[9], ÛÁ¡Ùé┤ Ù®öÝâÇÙÂäýäØ ýù░ÛÁ¼ Û▓░Û│╝ Û░äýä¼ý£áÝÖö ýèñý║öýùÉýä£ F2 ýºäÙï¿ýØÿ Û©░ýñÇý╣ÿÙèö 7.8 kPaýØ┤ýùêÙïñ[10]. Û░ä ýù╝ýªØ Ù░Å ýåÉýâü ýáòÙÅäÙÑ╝ ÝÅëÛ░ÇÝòá ÙòîÙèö Ýÿêý▓¡ ALTÛ░Ç ýú╝Ùí£ ÝÖ£ýÜ®ÙÉÿÙ®░, Û░äýâØÛ▓ÇýØä ÝåÁÝò┤ýä£ ýù╝ýªØ Û┤┤ýé¼ ÙÅÖÙ░ÿ ýù¼ÙÂÇÙÑ╝ ÝÖòýØ©Ýòá ýêÿ ý×êÙïñ. ýØ╝Ù░ÿýáüý£╝Ùí£ A2-A3 ýØ┤ýâüýØÿ ýù╝ýªØ ÙÅÖÙ░ÿ ýï£ ýñæÙô▒ÙÅä ýØ┤ýâüýØÿ ýù╝ýªØý£╝Ùí£ ÝîÉÙï¿Ýò£Ùïñ.
BÝÿòÛ░äýù╝ ý×Éýù░ Û▓¢Û│╝ ýñæ ýáäÝÿòýáüýØ© Û©░ýñÇýØÿ Ù®┤ýù¡Û┤Çýܮ۩░Ùèö HBeAg ýûæýä▒ýØ┤Û│á Ýÿêý▓¡ HBV DNA Û░ÆýØ┤ ÙîÇÛ░£ 107 IU/mL ýØ┤ýâüý£╝Ùí£ ÙºñýÜ░ ÙåÆýºÇÙºî, ALTÛ░Ç ýºÇýåìýáüý£╝Ùí£ ýáòýâü Ù▓öý£ä ýØ┤Ùé┤ýØ┤Ù®░ Û░äýâØÛ▓ÇýùÉýä£Ùèö ýù╝ýªØýØ┤ ýùåÛ▒░Ùéÿ ÙºñýÜ░ Û▓¢Ù»©ÝòÿÛ│á ýä¼ý£áÝÖöÙÅä ýùåÙèö ýâüÝâ£Ùí£, ýØ┤ ýï£Û©░ýùÉÙèö ý╣ÿÙúî ýùåýØ┤ Û▓¢Û│╝ Û┤Çý░░Ýòÿýù¼ÙÅä ýûæÝÿ©Ýò£ ýÿêÝøäÙÑ╝ Ù│┤ýØ©Ùïñ[11,12]. ÝòÿýºÇÙºî Ù®┤ýù¡Û┤Çýܮ۩░Ùí£ ýÂöýáòÙÉÿÙèö ÝÖÿý×ÉÙôñ ýñæýùÉýä£ÙÅä ýâüÙîÇýáüý£╝Ùí£ ÙéÿýØ┤Û░Ç ÙºÄýØÇ Û▓¢ýÜ░, Ùé¿ýä▒ýØ© Û▓¢ýÜ░, ýâüÙîÇýáüý£╝Ùí£ Ùé«ýØÇ Ýÿêý▓¡ HBV DNA ÙåìÙÅäÙÑ╝ Ù│┤ýØ┤Ùèö Û▓¢ýÜ░[13], Û░äýä¼ý£áÝÖö ýáÉýêÿÛ░Ç ÙåÆýØÇ Û▓¢ýÜ░[14], ALTÛ░Ç ýáòýâü ýâüÝò£ý╣ÿÙ│┤Ùïñ ýò¢Û░ä ÙåÆýØÇ Û▓¢ýÜ░ Ùô▒ýØ┤ Û░äýòö Ù░Å Û░ä Û┤ÇÙá¿ Ýò®Ù│æýªØ Ù░£ýâØ ý£äÝùÿýØ┤ ÙåÆýØÇ Û▓âý£╝Ùí£ Ù│┤Û│áÙÉÿýû┤ Ù®┤ýù¡Û┤Çýܮ۩░ ÝÜîýâëýºÇÙîÇ ÝÖÿý×ÉýùÉ ÙîÇÝò£ ýØ┤ýèêÛ░Ç ÙÂÇÛ░üÙÉÿýùêÙïñ. Ùö░ÙØ╝ýä£ ýØ┤Ù▓ê 2022Ùàä BÝÿòÛ░äýù╝ Û░ÇýØ┤Ùô£ÙØ╝ýØ©ýùÉýä£Ùèö Ù®┤ýù¡Û┤Çýܮ۩░ ýÂöýáò ÝÖÿý×É ýñæ ýáòýâü ALTÙÑ╝ Ù│┤ýØ┤ÙìöÙØ╝ÙÅä ýù░Ùá╣ýØ┤ 30-40ýä© ýØ┤ýâüýØ┤Û▒░Ùéÿ, Ýÿêý▓¡ HBV DNA < 107 IU/mLýØ© Û▓¢ýÜ░ ÙÿÉÙèö ALTÛ░Ç ýáòýâü ýâüÝò£ý╣ÿýØÿ Û▓¢Û│äýùÉ ý×êÙèö Û▓¢ýÜ░ýùÉÙèö Û░äýâØÛ▓Ç ÙÿÉÙèö Ù╣äý╣¿ýèÁýáü Ù░®Ù▓òý£╝Ùí£ Û░äýä¼ý£áÝÖö ýáòÙÅäÙÑ╝ ÝÅëÛ░ÇÝòÿýù¼ ýØÿÙ»© ý×êÙèö Û░äýä¼ý£áÝÖöÙÑ╝ ýï£ýé¼ÝòÿÙèö ýåîÛ▓¼ýØ┤ ý×êýØä Û▓¢ýÜ░, ý╣ÿÙúîÙÑ╝ Û│áÙáñÝòá ýêÿ ý×êýØîýØä ÛÂîÛ│áÝòÿýÿÇÙïñ(ÛÂîÛ│á ýêÿýñÇ: B2) (Fig. 1).
Ùºîýä▒ BÝÿòÛ░äýù╝ ÝÖÿý×É ýñæ Ù░öýØ┤Ùƒ¼ýèñýØÿ ÝÖ£ÙÅÖýáü ýªØýïØÛ│╝ Ýò¿Û╗ÿ ýñæÙô▒ÙÅä ýØ┤ýâüýØÿ ýù╝ýªØÛ│╝ 2Ùï¿Û│ä ýØ┤ýâüýØÿ ýØÿÙ»© ý×êÙèö ýä¼ý£áÝÖöÙÑ╝ Ù│┤ýØ┤Ùèö Û▓¢ýÜ░ Ù®┤ýù¡ÝÖ£ÙÅÖÛ©░Ùí£ ýáòýØÿÝò£Ùïñ. Ù░öýØ┤Ùƒ¼ýèñýØÿ ÝÖ£ÙÅÖýä▒ ýªØýïØýØÇ HBV DNAÙí£ ÝÅëÛ░ÇÝòÿÙ®░ HBeAg ýûæýä▒ýØÿ Û▓¢ýÜ░ýùÉÙèö 2,000-20,000 IU/mL ýØ┤ýâü, HBeAg ýØîýä▒ýØ© Û▓¢ýÜ░ýùÉÙèö 2,000 IU/mL ýØ┤ýâüýØ© Û▓¢ýÜ░ÙÑ╝ Ýò¡Ù░öýØ┤Ùƒ¼ýèñ ý╣ÿÙúîÙÑ╝ Û│áÙáñÝòÿÙèö Ù░öýØ┤Ùƒ¼ýèñ ÝÖ£ÙÅÖýáü ýªØýïØý£╝Ùí£ Û░äýú╝Ýò£Ùïñ. Û░äýØÿ ýù╝ýªØýØ┤ ý×êÙèöýºÇ ÝÖòýØ©ÝòÿÙèö Ùì░Ùèö Ýÿêý▓¡ ALTÛ░Ç Û░äýåÉýâüýØä Ù░ÿýÿüÝòÿÙèö Û░äÝÄ©Ýò£ ýºÇÝæ£Ùí£ ÙäÉÙª¼ ÝÖ£ýÜ®ÙÉÿýû┤ ýÖöÙïñ. ýÁ£ÛÀ╝ 15Û░£ýØÿ Ù¼┤ý×æý£ä ÙîÇýí░ÛÁ░ ýù░ÛÁ¼ýÖÇ 44Û░£ýØÿ Û┤Çý░░ ýù░ÛÁ¼ÙÑ╝ ÙÂäýäØÝò£ Ù®öÝâÇÙÂäýäØýùÉýä£ÙÅä Ù®┤ýù¡ÝÖ£ÙÅÖÛ©░ ÝÖÿý×ÉÛÁ░ýùÉýä£ýØÿ Ýò¡Ù░öýØ┤Ùƒ¼ýèñ ý╣ÿÙúîÙèö Û░äÛ▓¢Ù│ÇýªØýØÿ ý£äÝùÿ, Ù╣äÙîÇýâüýä▒ Ù│ÇÝÖöýØÿ ý£äÝùÿ ÛÀ©Ùª¼Û│á Û░äýòöýØÿ ý£äÝùÿýØä Ùé«ýÂöÙèö Û▓âý£╝Ùí£ ÙéÿÝâÇÙé¼Ùïñ[15]. Ùö░ÙØ╝ýä£ Ù░öýØ┤Ùƒ¼ýèñýØÿ ÝÖ£ÙÅÖýáü ýªØýïØÛ│╝ ÙìöÙÂêýû┤ ýºäÝûëÙÉ£ ýù╝ýªØ Ù░Å ýä¼ý£áÝÖöÙÑ╝ Ù│┤ýØ┤Ùèö Û▓¢ýÜ░ýùÉÙèö Ýò¡Ù░öýØ┤Ùƒ¼ýèñ ý╣ÿÙúîÙÑ╝ ýï£ý×æÝò┤ýò╝ Ýò£Ùïñ(Fig. 1).
HBV DNAÙèö ýªØÛ░ÇÙÉÿýû┤ ý×êý£╝Ùéÿ ALTÛ░Ç ýáòýâü ýâüÝò£ý╣ÿýØÿ 1-2Ù░░ ýé¼ýØ┤Ùí£ ýºÇýåìÙÉÿýû┤ Ù®┤ýù¡ÝÖ£ÙÅÖÛ©░ýØÿ Û©░ýñÇýùÉ ÙÂÇÝò®ÝòÿýºÇ ýòèÙèö ÝÜîýâë ýºÇÙîÇýØÿ Û▓¢ýÜ░, 2022Ùàä Û░£ýáò Û░ÇýØ┤Ùô£ÙØ╝ýØ©ýùÉýä£Ùèö ýÂöýáü Û┤Çý░░ÝòÿÛ▒░Ùéÿ Û░äýâØÛ▓ÇýØä ýï£ÝûëÝòÿýù¼ ýñæÙô▒ÙÅä ýØ┤ýâüýØÿ ýù╝ýªØ Û┤┤ýé¼ Ýÿ╣ýØÇ Ù¼©ÙºÑ ýú╝Ù│ÇÙÂÇ ýä¼ý£áÝÖö ýØ┤ýâüýØÿ Ùï¿Û│äÙÑ╝ Ù│┤ýØ┤Ù®┤ Ýò¡Ù░öýØ┤Ùƒ¼ýèñ ý╣ÿÙúîÙÑ╝ ýï£ý×æÝòÿÙÅäÙíØ ÛÂîÛ│áÝòÿýÿÇÙïñ(ÛÂîÛ│á ýêÿýñÇ: A1). ÙÿÉÝò£ ý×äýâüýáüý£╝Ùí£ ýØÿÙ»©ý×êÙèö Û░äýä¼ý£áÝÖöÙèö Ù╣äý╣¿ýèÁýáü Ù░®Ù▓òý£╝Ùí£ ÝÅëÛ░ÇÝòá ýêÿ ý×êÙÅäÙíØ ýá£ýï£ÝòÿýÿÇÙïñ(ÛÂîÛ│á ýêÿýñÇ: B1) (Fig. 1).
ýáäÝÿòýáüýØ© Ù®┤ýù¡Ù╣äÝÖ£ÙÅÖÛ©░Ùèö HBeAg ýØîýä▒, anti-HBe ýûæýä▒, ýºÇýåìýáüýØ© ýáòýâü ALT ÛÀ©Ùª¼Û│á HBV DNAÛ░Ç ý©íýáòý╣ÿ ýØ┤ÝòÿÙí£ Ù»©Û▓Çý£ÙÉÿÛ▒░Ùéÿ, Ùé«ýØÇ ÙåìÙÅä(< 2 ,000 IU/mL)Ùí£ ýºÇýåìÙÉÿÙèö ýï£Û©░Ùí£ ýºäÝûëÙÉ£ Û░äýä¼ý£áÝÖöýØÿ ýªØÛ▒░Û░Ç ýùåÙèö Ù®┤ýù¡Ù╣äÝÖ£ÙÅÖÛ©░ýØÿ Û▓¢ýÜ░ ý╣ÿÙúîýùåýØ┤ Û▓¢Û│╝ Û┤Çý░░Ýòÿýù¼ÙÅä ýóïýØÇ ýÿêÝøäÙÑ╝ Ù│┤ýØ©Ùïñ[16]. Ù░ÿÙ®┤ Ùé«ýØÇ ÙåìÙÅäýØÿ Ù░öýØ┤Ùƒ¼ýèñ Ýÿêýñæ ýâüÝâ£ÙÑ╝ Ù│┤ýØ┤ÙìöÙØ╝ÙÅä(< 2 ,000 IU/mL), ýºäÝûëÙÉ£ Û░äýä¼ý£áÝÖöÙÑ╝ ýï£ýé¼ÝòÿÙèö FIB-4Ùéÿ APRIÛ░Ç ýªØÛ░ÇÝò£ Û▓¢ýÜ░ Û░äýòö Ùô▒ Ýò®Ù│æýªØ Ù░£ýâØýØ┤ ýªØÛ░ÇÙÉÿýû┤ ý×êÙèö Û▓âý£╝Ùí£ Ù│┤Û│áÙÉÿýùêÙïñ[5,17,18]. Ùö░ÙØ╝ýä£ Û░£ýáòÙÉ£ Û░ÇýØ┤Ùô£ÙØ╝ýØ©ýùÉýä£Ùèö Ýÿêý▓¡ HBV DNA < 2,000 IU/mLÙØ╝ÙÅä Û░äýâØÛ▓ÇýØ┤Ùéÿ Ù╣äý╣¿ýèÁýáü Û▓Çýé¼ýùÉýä£ ýØÿÙ»© ý×êÙèö Û░äýä¼ý£áÝÖöÙÑ╝ ýï£ýé¼ÝòÿÙèö ýåîÛ▓¼ýØ┤ ý×êýØä Û▓¢ýÜ░, ý╣ÿÙúîÙÑ╝ Û│áÙáñÝòÿÙÅäÙíØ ÝòÿýÿÇÙïñ(ÛÂîÛ│á ýêÿýñÇ: B2) (Fig. 1).
Û░äÛ▓¢Ù│ÇýªØýØÿ Ýò®Ù│æýªØýØ┤ Ù░£ýâØÝòÿýºÇ ýòèýØÇ ÙîÇýâüýä▒ Û░äÛ▓¢Ù│ÇýªØ ÝÖÿý×ÉýØÿ Û▓¢ýÜ░ Ýÿêý▓¡ HBV DNA ÔëÑ 2,000 IU/mLýØ┤Ù®┤ ALTýùÉ Û┤ÇÛ│äýùåýØ┤ Ýò¡Ù░öýØ┤Ùƒ¼ýèñýᣠý╣ÿÙúîÛ░Ç Û©ëýù¼ ýáüýÜ®ýØ┤ Û░ÇÙèÑÝòÿÙïñ. ýÁ£ÛÀ╝ ý£áÙƒ¢ Ù░Å Ù»©ÛÁ¡ Û░äÝòÖÝÜî Û░ÇýØ┤Ùô£ÙØ╝ýØ©ýùÉýä£Ùèö Ùé«ýØÇ ÙåìÙÅäýØÿ Ýÿêý▓¡ HBV DNA ÙåìÙÅäÙÑ╝ Ù│┤ýØ┤ÙìöÙØ╝ÙÅä(< 2 ,000 IU/mL) Ýÿêý▓¡ HBV DNAÛ░Ç Û▓Çý£ÙÉÿÙèö ÙîÇýâüýä▒ Û░äÛ▓¢Ù│ÇýªØ ÝÖÿý×ÉýùÉýä£ ý╣ÿÙúîÙÑ╝ ÛÂîÛ│áÝòÿÛ│á ý×êÙïñ[11,19]. Ùé«ýØÇ Ýÿêý▓¡ HBV DNA ÙåìÙÅäÙÑ╝ Ù│┤ýØ┤Ùèö ÙîÇýâüýä▒ Û░äÛ▓¢Ù│ÇýªØ ÝÖÿý×ÉÙôñýØÿ ý×äýâü Û▓¢Û│╝ÙÑ╝ Ù│┤Û│áÝò£ ÛÁ¡Ùé┤ Û┤Çý░░ ýù░ÛÁ¼ýùÉýä£Ùèö Û┤Çý░░ Û©░Û░ä ýñæ 33%Û░Ç HBV DNA 2,000 IU/mL ýØ┤ýâü ýâüýè╣ ýåîÛ▓¼ýØä Ù│┤ýÿÇÛ│á, ýØ┤Ùôñ ýñæýùÉýä£ Û░äýòöýØÿ ý£äÝùÿýØ┤ ÙåÆÙïñÛ│á Ù│┤Û│áÝòÿýÿÇÙïñ[20]. ÙÿÉÝò£ Ù╣äÙíØ Û┤Çý░░ Û©░Û░ä Ùé┤Ùé┤ Ùé«ýØÇ ÙåìÙÅäýØÿ Ù░öýØ┤Ùƒ¼ýèñÙí£ ý£áýºÇÙÉ£ÙïñÛ│á ÝòÿÙìöÙØ╝ÙÅä Ù░öýØ┤Ùƒ¼ýèñÛ░Ç Ù»©Û▓Çý£ ýâüÝâ£Ùí£ ý£áýºÇÙÉ£ ÝÖÿý×ÉÛÁ░ýùÉ Ù╣äÝòÿýù¼ Û░äýòöýØÿ ý£äÝùÿýØ┤ ÙåÆýòÿÙïñ. Ùö░ÙØ╝ýä£ Ùé«ýØÇ Ýÿêý▓¡ HBV DNA ÙåìÙÅäÙÑ╝ Ù│┤ýØ┤Ùèö Û░äÛ▓¢Ù│ÇýªØ ÝÖÿý×ÉýùÉÛ▓î Ýò¡Ù░öýØ┤Ùƒ¼ýèñ ý╣ÿÙúîÙÑ╝ ýï£ÝûëÝòÿÛ▓î ÙÉÿÙ®┤ HBV DNAýØÿ ÙåìÙÅä ýâüýè╣ Ù░£ýâØýØä ýÿêÙ░®Ýòá ýêÿ ý×êÛ│á, Ýò®Ù│æýªØ Ù░£ýâØýØÿ ý£äÝùÿýØä Ùé«ý£ Û░ÇÙèÑýä▒ýØ┤ ý×êÙïñ. ýØ┤ÙÑ╝ Û©░Ù░ÿý£╝Ùí£ Û░£ýáò Û░ÇýØ┤Ùô£ÙØ╝ýØ©ýùÉýä£Ùèö Ýÿêý▓¡ HBV DNA < 2,000 IU/mLÙØ╝ÙÅä Ýÿêý▓¡ HBV DNAÛ░Ç Û▓Çý£ÙÉÿÙèö ÙîÇýâüýä▒ Û░äÛ▓¢Ù│ÇýªØýØÿ Û▓¢ýÜ░ýùÉÙèö ALTýùÉ Û┤ÇÛ│äýùåýØ┤ Ýò¡Ù░öýØ┤Ùƒ¼ýèñ ý╣ÿÙúîÙÑ╝ ýï£ý×æÝòá Û▓âýØä ÛÂîÛ│áÝòÿýÿÇÙïñ(ÛÂîÛ│á ýêÿýñÇ: B1) (Fig. 1).
Ùºîýä▒ BÝÿòÛ░äýù╝ ÝÖÿý×ÉýùÉýä£ ý╣ÿÙúîýØÿ ýóàÙúîÙÑ╝ Û│áÙáñÝòÿÛ│áý×É Ýò£ÙïñÙ®┤ ý╣ÿÙúî Ù¬®ýáüýØä ý×ÿ Ù░ÿýÿüÝòá ýêÿ ý×êý£╝Ù®┤ýä£ ý╣ÿÙúî ýñæ ý©íýáòýØ┤ ýÜ®ýØ┤Ýò£ ÙîÇý▓┤ ýºÇÝæ£ýØÿ Ù│ÇÝÖöÙÑ╝ ý╣ÿÙúî Ù¬®Ýæ£Ùí£ ýé╝ýòä ýóàÙúî ýï£ýáÉýØä ýäáÝâØÝòÿÙèö Û▓âýØ┤ ÙîÇýòêýØ┤ ÙÉá ýêÿ ý×êÙïñ. ý×äýâüýùÉýä£Ùèö ALT ýáòýâüÝÖö, Ýÿêý▓¡ HBV DNAýØÿ ÙÂêÛ▓Çý£, HBeAgýØÿ Ýÿêý▓¡ýåîýïñ Ù░Å ýáäÝÖÿ, HBsAg Ýÿêý▓¡ýåîýïñ Ù░Å ýáäÝÖÿ Ùô▒ýØä ý╣ÿÙúî Ù¬®Ýæ£Ùí£ ýØ┤ýÜ®ÝòÿÛ│á ý×êÙïñ. ýÁ£ÛÀ╝, Ýÿêý▓¡ HBsAg ýáòÙƒë Û▓Çýé¼, Ýÿêý▓¡ hepatitis B core-related antigen (HBcrAg) ýáòÙƒë Û▓Çýé¼ Ýÿ╣ýØÇ Ýÿêý▓¡ HBV RNA Ùô▒ýØÿ ý©íýáòýØä ÝåÁÝò┤ýä£ ýºÇýåì ý╣ÿÙúîÙ░ÿýØæ ýÿêý©í Ù░Å ýóàÙúî ýï£ýáÉ Û▓░ýáòýùÉ ÙÅäýøÇýØä ýñä ýêÿ ý×êÙïñÙèö ýù░ÛÁ¼ÙôñýØ┤ ýåîÛ░£ ÙÉÿÛ│á ý×êÙïñ.
Ýò¡Ù░öýØ┤Ùƒ¼ýèñ ý╣ÿÙúî Ýøä ý╣ÿÙúî Ù░ÿýØæýØ┤ ýºÇýåìÙÉÿÛ©░ÙÅä ÝòÿýºÇÙºî ýò¢ýᣠýñæÙï¿ Ýøä Û░äýù╝ ýòàÝÖö Ù░Å ýï¼Û░üÝò£ Û░äÙÂÇýáäý£╝Ùí£ ýºäÝûëÝòá ýêÿ ý×êÙïñ. Ùö░ÙØ╝ýä£ ý╣ÿÙúî Ù░ÿýØæ ýºÇýåì Ù░Å ý×¼Ù░£ ýù¼ÙÂÇ, Û░äÛ©░ÙèÑ ýâüÝ⣠Ùô▒ýØä ýòîýòäÙ│┤Û©░ ý£äÝòÿýù¼ ýáòÛ©░ýáüýØ© Û░äÛ©░ÙèÑ Û▓Çýé¼, HBeAg, anti-HBe, HBV DNA Ùô▒ýùÉ ÙîÇÝò£ ýÂöýáü Û▓Çýé¼ Ù░Å Û▓¢Û│╝ Û┤Çý░░ýØ┤ Ù░ÿÙô£ýï£ ÝòäýÜöÝòÿÙïñ. Ýè╣Ý×ê, ýò¢ýᣠýñæÙï¿ Ýøä HBV DNAÛ░Ç ýªØÛ░ÇÝòÿÙèö Û▓¢ýÜ░, HBV DNAýÖÇ ALTÙÑ╝ ÝżÝò¿Ýò£ Û░äÛ©░ÙèÑ Û▓Çýé¼ÙÑ╝ ýä©Ù░ÇÝ×ê ýï£ÝûëÝòÿýù¼ ýò¢ýᣠý×¼Ýê¼ýù¼ ýù¼ÙÂÇÙÑ╝ Û▓░ýáòÝò┤ýò╝ Ýò£Ùïñ.
HBsAg ýåîýïñýØÇ anti-HBs Ýÿòýä▒ ýù¼ÙÂÇýÖÇ ýâüÛ┤ÇýùåýØ┤ Ýÿêýòí Ùé┤ýØÿ HBsAgÛ│╝ HBV DNAýØÿ ýåîýïñýØä Ù£╗Ýò£Ùïñ[21]. ýØ┤Ùƒ¼Ýò£ Û▓¢ýÜ░ Ù╣äÙíØ Û░äýä©Ýż Ùé┤ýùÉ cccDNAÛ░Ç ýí┤ý×¼Ýòá ýêÿ ý×êýºÇÙºî Û░äýåÉýâüýØ┤ ýºäÝûëÙÉÿýºÇ ýòèÛ│á Û░äýòöýØÿ ý£äÝùÿýØ┤ Ùé«ýòä ÔÇ£Û©░ÙèÑýáü ýÖäý╣ÿÔÇØÙí£ ýØ╝ý╗¼ýû┤ýºÇÙ®░ ÝÿäýïñýáüýØ© ý╣ÿÙúî Ù¬®Ýæ£Ùí£ ýá£ýï£ÙÉÿÛ│á ý×êÙïñ. Ýò¡Ù░öýØ┤Ùƒ¼ýèñ ý╣ÿÙúîÙÑ╝ ÝåÁÝòÿýù¼ HBsAg ýåîýïñ Ýÿ╣ýØÇ ýáäÝÖÿýØ┤ ýØ╝ýû┤ÙéÿÙèö ÝÖÿý×ÉýùÉýä£ ýØ╝ÙÂÇÛ░Ç ýØ╝ýï£ýáüýØ© HBsAg ý×¼ý£ÝÿäýØ┤Ùéÿ HBV DNAýØÿ Û▓Çý£ýØ┤ ÙÉá ýêÿ ý×êý£╝Ùéÿ ÙîÇÙÂÇÙÂäýØÿ ÝÖÿý×ÉÙèö HBsAg ýåîýïñÛ│╝ HBV DNA ÙÂêÛ▓Çý£ýØä ý£áýºÇÝò£ÙïñÛ│á Ù│┤Û│áÙÉÿýùêÛ│á Û░äýòöýØÿ Ù░£ýâØÙÅä HBsAg ýåîýïñýØä Ù│┤ýØ┤ýºÇ ýòèýØÇ ÝÖÿý×ÉýùÉ Ù╣äÝòÿýù¼ Ùé«ý£╝Ù»ÇÙí£, HBsAg ýåîýïñ Ýÿ╣ýØÇ ýáäÝÖÿýØÇ ý╣ÿÙúî Ù¬®ýáüýØä ý×ÿ Ù░ÿýÿüÝòÿÙèö Û░Çý×Ñ ýóïýØÇ ý×äýâüýáüýØ© ý╣ÿÙúî Ù¬®Ýæ£ÙØ╝ Ýòá ýêÿ ý×êÙïñ[22-24]. Ùö░ÙØ╝ýä£, Ùºîýä▒ BÝÿòÛ░äýù╝ ÝÖÿý×ÉýùÉýä£Ùèö HBsAg ýåîýïñýØ┤ ýØ┤Ùú¿ýû┤ýºä Ýøä Û▓¢ÛÁ¼ýÜ® Ýò¡Ù░öýØ┤Ùƒ¼ýèñýᣠý╣ÿÙúî ýóàÙúîÙÑ╝ ÛÂîý×ÑÝò£Ùïñ(ÛÂîÛ│á ýêÿýñÇ: A1).
HBVýØÿ Ù│ÁýᣠÛ│╝ýáòýùÉýä£ ýáäýé¼ÙÉ£ mRNAÙí£ÙÂÇÝä░ ýä© Û░ÇýºÇ ÙïñÙÑ© Ýü¼Û©░ýØÿ S (small), M (middle), L (large) HBs Ùï¿Ù░▒ýØ┤ ýâØýä▒ýØ┤ ÙÉÿÙèöÙì░, HBsAg ýáòÙƒëý╣ÿÙèö ýØ┤ ýä© Û░ÇýºÇ ý×àý×É Ù¬¿ÙæÉÙÑ╝ ÝżÝò¿Ýòÿýù¼ Û▓Çý£ÝòÿÛ▓î ÙÉ£Ùïñ[25]. ýù¼Ùƒ¼ ýù░ÛÁ¼ýùÉýä£ Ýò¡Ù░öýØ┤Ùƒ¼ýèñýᣠý╣ÿÙúî ýóàÙúî ýï£ýáÉýØÿ Ùé«ýØÇ HBsAg ýáòÙƒëý╣ÿÛ░Ç ýºÇýåì Ù░öýØ┤Ùƒ¼ýèñ Ù░ÿýØæÛ│╝ HBsAg Ýÿêý▓¡ ýåîýïñýØä ýÿêý©íÝòÿÙèöÙì░ ý£áýÜ®Ýò¿ýØ┤ Ù│┤Û│áÙÉÿýùêÙïñ[26,27]. ý╣ÿÙúî ýóàÙúî ýï£ýáÉýùÉ 10-200 IU/mL ýáòÙÅäýØÿ Ùé«ýØÇ HBsAg ýáòÙƒëý╣ÿÙÑ╝ Ù│┤ýØ┤Ùèö Û▓¢ýÜ░ ýò¢ýᣠýóàÙúî ÝøäýùÉÙÅä ýºÇýåì Ù░öýØ┤Ùƒ¼ýèñ Ù░ÿýØæÛ│╝ HBsAg ýåîýïñÛ│╝ ýù░Û┤ÇÙÉ£ÙïñÛ│á ýòîÙáñýá© ý×êýû┤, Û▓¢ÛÁ¼ýÜ® Ýò¡Ù░öýØ┤Ùƒ¼ýèñýᣠý╣ÿÙúî ýñæ Ù¬¿ÙïêÝä░ÙºüýùÉ ý×êýû┤ýä£ÙÅä HBsAg ýáòÙƒë Û▓Çýé¼Ùèö ÙÅäýøÇýØ┤ ÙÉ£Ùïñ. Ùö░ÙØ╝ýä£ 2022Ùàä Û░£ýáò Û░ÇýØ┤Ùô£ÙØ╝ýØ©ýùÉýä£Ùèö Û▓¢ÛÁ¼ýÜ® Ýò¡Ù░öýØ┤Ùƒ¼ýèñýᣠý╣ÿÙúî ýñæÙï¿ýØä Û│áÙáñÝòÿÙèö Û▓¢ýÜ░, HBsAg ýáòÙƒëý╣ÿÙÑ╝ ý░©Û│áÝòÿýù¼ ýñæÙï¿ ýù¼ÙÂÇÙÑ╝ Û▓░ýáòÝòá ýêÿ ý×êÙÅäÙíØ ÛÂîÛ│áÝòÿýÿÇÙïñ(ÛÂîÛ│á ýêÿýñÇ: B1).
HBcrAgýØÇ precore/core ý£áýáäý×ÉÙí£ÙÂÇÝä░ Ýò®ýä▒ÙÉ£ 3Û░£ýØÿ Ùï¿Ù░▒ýºê(HBeAg, HBcAg, p22cr)ýØä ÙÅÖýï£ýùÉ ý©íýáòÝòÿÙèö Ýÿêý▓¡Ýæ£ýºÇý×ÉýØ┤Ùïñ. HBcrAgýØÇ Û░ä Ùé┤ Ù░öýØ┤Ùƒ¼ýèñ ýªØýïØ ýáòÙÅäÙÑ╝ ÙéÿÝâÇÙé┤Ùèö Û░äÙé┤ cccDNA, Û░äÙé┤ HBV DNAýÖÇ ýóïýØÇ ýâüÛ┤ÇÛ┤ÇÛ│äÙÑ╝ Ù│┤ýØ┤Ùèö Û▓âý£╝Ùí£ ýòîÙáñýá© ý×êÙïñ[25,28]. HBcrAg ýáòÙƒëý╣ÿÙÅä Ýò¡Ù░öýØ┤Ùƒ¼ýèñýᣠý╣ÿÙúî Ýøä ýºÇýåì Ù░ÿýØæýØä ýÿêý©íÝòÿÙèö Ùì░ ý£áýÜ®ÝòÿÙ®░, ýÁ£ÛÀ╝ ýØ╝Ù│© Û░ÇýØ┤Ùô£ÙØ╝ýØ©ýùÉýä£Ùèö HBsAg ýáòÙƒëý╣ÿýÖÇ HBcrAg ýáòÙƒëý╣ÿÙÑ╝ ýí░Ýò®Ýòÿýù¼ ý×¼ÝÖ£ýä▒ÝÖö ý£äÝùÿÙÅäýùÉ Ùö░ÙØ╝ ýò¢ýᣠýñæÙï¿ýØä ÛÂîÛ│áÝòÿÛ│á ý×êÙïñ[29].
Ýÿêý▓¡ HBV RNAÙèö cccDNAÙí£ÙÂÇÝä░ ýºÇýåìýáüý£╝Ùí£ ýáäýé¼ÙÉÿýû┤ ýâØýä▒ÙÉÿÙ®░, Ýÿêý▓¡ HBV DNA, HBsAgÛ│╝ ÙìöÙÂêýû┤ Û░äýùÉýä£ ýâØýä▒ÙÉÿÙèö Ù░öýØ┤Ùƒ¼ýèñ ýªØýïØ ýñæÛ░äÙ¼╝(viral replicative intermediates)ýØä ÙîÇÙ│ÇÝòÿÙèö Ýÿêý▓¡ ýºÇÝæ£ýØ┤Ùïñ[30]. HBV RNAÙÅä ýÁ£ÛÀ╝ Ýò¡Ù░öýØ┤Ùƒ¼ýèñýᣠý╣ÿÙúî ýóàÙúîÙÑ╝ Û▓░ýáòÝòÿÙèö ýØ©ý×ÉÙí£ ýù░ÛÁ¼ÙôñýØ┤ ýºäÝûëÙÉÿÛ│á ý×êÙïñ[7,31]. ÙÿÉÝò£, ýØ┤ÙÑ╝ HBsAg ýáòÙƒëý╣ÿýÖÇ HBcrAg ýáòÙƒëý╣ÿ Ùô▒Û│╝ýØÿ ýí░Ýò®ý£╝Ùí£ ý╣ÿÙúî ýóàÙúî ÝøäýØÿ ýºÇýåì Ù░öýØ┤Ùƒ¼ýèñ Ù░ÿýØæýØä ýÿêý©íÝòÿÙèö ýºÇÝæ£Ùí£ ýá£ýï£ÝòÿÛ│á ý×êÙïñ[32,33]. ÛÀ©Ùƒ¼Ùéÿ Û░ÖýØÇ ÝÖÿý×ÉýØÿ Ýÿêý▓¡ýùÉýä£ÙÅä HBV RNA ýÂöý£ Ù░Å PCR ýáòÙƒë Ù░®Ù▓òýùÉ Ùö░ÙØ╝ Û▓░Û│╝Û░ÆýØ┤ Ùï¼ÙØ╝ýºÇÙ»ÇÙí£, HBV RNAÛ░Ç BÝÿòÛ░äýù╝ýØÿ ý£áýÜ®Ýò£ Ýæ£ýºÇý×ÉÙí£ ÙäÉÙª¼ ýé¼ýÜ®ÙÉÿÛ©░ ý£äÝò┤ýä£Ùèö ÝûÑÝøä HBV RNA Û▓Çý£ Ù░®Ù▓òýØÿ Ýæ£ýñÇÝÖöÛ░Ç ÝòäýÜöÝòÿÙïñ.
BÝÿòÛ░äýù╝ýùÉ ýØÿÝò┤ Û░äýòöýØ┤ Ù░£ýâØÝò£ ÝÖÿý×ÉýùÉýä£ Ýò¡Ù░öýØ┤Ùƒ¼ýèñýᣠý╣ÿÙúîÙèö BÝÿòÛ░äýù╝ Ù░öýØ┤Ùƒ¼ýèñ ýªØýïØýØä ýûÁýá£Ýòÿýù¼ Û░äÛ©░ÙèÑýØä Ýÿ©ýáäýï£ý╝£ Û░äýòöýùÉ ÙîÇÝò┤ ýáüÛÀ╣ýáüýØ© ý╣ÿÙúîÙÑ╝ Ù░øýØä ýêÿ ý×êÙÅäÙíØ ÙÅäýÖÇýú╝Ùèö ýù¡ÝòáýØä Ýò£Ùïñ. ÙÿÉÝò£ Û░äýòöýùÉ ÙîÇÝò┤ Û░ä ýáêýá£, Û│áýú╝Ýîî ýù┤ý╣ÿÙúî, Û░äýØ┤ýïØ Ùô▒ýØÿ ýÖäý╣ÿ ý╣ÿÙúîÙÑ╝ Ù░øýØÇ ÝÖÿý×ÉýùÉýä£ Ýò¡Ù░öýØ┤Ùƒ¼ýèñýᣠý╣ÿÙúîÙèö Û░äýòöýØÿ ý×¼Ù░£ ý£äÝùÿýØä ýñäýØ┤Û│á ýâØýí┤ý£¿ýØä ÝûÑýâüýï£ÝéñÙèö Û▓âý£╝Ùí£ ýòîÙáñýíîÙïñ[33,34]. ýØ┤Ùƒ¼Ýò£ ýáÉýØä Û│áÙáñÝòÿýù¼ ÛÁ¡Ùé┤ýùÉýä£Ùèö BÝÿòÛ░äýù╝ ÝÖÿý×ÉÛ░Ç Û░äýòöýØä ýºäÙï¿Ù░øÙèö Û▓¢ýÜ░, HBV DNAÛ░Ç Û▓Çý£ÙÉÿÛ©░Ùºî ÝòÿÙ®┤ ALT, AST ýáòÙÅäýÖÇ ýâüÛ┤ÇýùåýØ┤ Ýò¡Ù░öýØ┤Ùƒ¼ýèñýᣠýé¼ýÜ®ýØä ÛÂîÛ│áÝòÿÛ│á ý×êý£╝Ù®░ Ù│┤Ýùÿ ýáüýÜ®ÙÅä Û░ÇÙèÑÝòÿÙïñ. Ýò¡Ù░öýØ┤Ùƒ¼ýèñýᣠýóàÙÑÿýØÿ Û▓¢ýÜ░ ÝàîÙà©ÝżÙ╣äýû┤, ýùöÝàîý╣┤Ù╣äýû┤ Ùô▒ýØÿ Ù░öýØ┤Ùƒ¼ýèñ ýûÁýá£ÙèÑýØ┤ ÙåÆýØÇ ýò¢ýá£ÙÑ╝ ýé¼ýÜ®ÝòÿÙèö Û▓âýØ┤ ÛÀ©ÙáçýºÇ ýòèýØÇ Û▓¢ýÜ░Ù│┤Ùïñ Û░äýòö ý╣ÿÙúî Ýøä Ù¼┤ý×¼Ù░£ ýâØýí┤ Û©░Û░äýØ┤ ý£áýØÿÝòÿÛ▓î Û©©ýùêÙïñ. ÝòÿýºÇÙºî ÝàîÙà©ÝżÙ╣äýû┤ýÖÇ ýùöÝàîý╣┤Ù╣äýû┤ ýñæ ýû┤ÙèÉ ýò¢ýá£Û░Ç Û░äýòö ý×¼Ù░£ Ù░Å ýâØýí┤ý£¿ ÝûÑýâüýùÉ Ùìö ÝÜ¿Û│╝ýáüýØ©ýºÇýùÉýä£Ùèö ýù¼Ùƒ¼ ÝøäÝûÑýáü ýù░ÛÁ¼ýùÉýä£ ýâüÙ░ÿÙÉ£ Û▓░Û│╝ÙÑ╝ Ù│┤ýù¼ ýÂöÛ░ÇýáüýØ© ÙîÇÛÀ£Ù¬¿ ýù░ÛÁ¼ÙôñýØ┤ ÝòäýÜöÝòÿÛ▓áÙïñ[35,36].
Û░äýòö ÝÖÿý×ÉÛ░Ç BÝÿòÛ░äýù╝ Ýæ£Ù®┤Ýò¡ýøÉ(HBsAg)ýØÇ ýûæýä▒ýØ┤Ùéÿ HBV DNAÛ░Ç Û▓Çý£ÙÉÿýºÇ ýòèÙèö Û▓¢ýÜ░ Ýÿäý×¼Ùèö Ýò¡Ù░öýØ┤Ùƒ¼ýèñýá£Û░Ç Ù│┤Ýùÿ ýáüýÜ®ýØ┤ ÙÉÿýºÇ ýòèÙèöÙïñ. ÝòÿýºÇÙºî ýØ┤Ùƒ░ Û▓¢ýÜ░ýùÉÙÅä ýêÿýêáýáü ýáêýᣠÝøä HBV ý×¼ÝÖ£ýä▒ÝÖöÛ░Ç 20% Û░ÇÛ╣îýØ┤ Ù░£ýâØÝò£ Ù░ÿÙ®┤, Ýò¡Ù░öýØ┤Ùƒ¼ýèñýᣠýé¼ýÜ®ÝòÿÙèö ÛÁ░ýùÉýä£Ùèö HBV ý×¼ÝÖ£ýä▒ÝÖöÙÑ╝ Û░Éýåîýï£ý╝░Ùïñ[37]. Ùö░ÙØ╝ýä£ HBV DNAÛ░Ç Û▓Çý£ÙÉÿýºÇ ýòèÙìöÙØ╝ÙÅä HBsAg ýûæýä▒ýØ© Û░äýòö ÝÖÿý×ÉÙôñýØÇ Ýò¡Ù░öýØ┤Ùƒ¼ýèñýᣠýé¼ýÜ®ýØä Û│áÙáñÝòÿÙèö Û▓âýØ┤ ýóïÛ▓áÙïñ.
Û░äýòö ý╣ÿÙúî ýñæ Û▓¢ÙÅÖÙºÑÝò¡ýòöÝÖöÝòÖýâëýáäýêá(transarterial chemoembolization, TACE)ýÖÇ ý▓┤ýÖ© Ù░®ýé¼ýäá ý╣ÿÙúîÙÑ╝ Ù│æÝûëÝòá Û▓¢ýÜ░ýùÉÙèö Ùï¿ÙÅà TACE ý╣ÿÙúîýùÉ Ù╣äÝò┤ HBV ý×¼ÝÖ£ýä▒ÝÖö ý£äÝùÿýØ┤ 2Ù░░ ýØ┤ýâü ýªØÛ░ÇÝòÿýÿÇÙïñ[38]. ÙÿÉÝò£ HBsAg ýØîýä▒ýØ┤ýºÇÙºî anti-HBc ýûæýä▒ýØ┤Û▒░Ùéÿ Û│╝Û▒░ BÝÿòÛ░äýù╝ýØÿ Ù│æÙáÑýØ┤ ý×êýùêÙìÿ ÝÖÿý×ÉýùÉýä£ÙÅä TACE Ùï¿ÙÅà ý╣ÿÙúîÙéÿ TACEýÖÇ ý▓┤ýÖ© Ù░®ýé¼ýäá ý╣ÿÙúî Ù│æÝûë ýï£ HBV ý×¼ýûæýáä Ù░Å ý×¼ÝÖ£ýä▒ÝÖöÛ░Ç ÙÉ£ÙïñÙèö ýù░ÛÁ¼Û░Ç Ù│┤Û│áÙÉÿýùêÙïñ[39]. Ùö░ÙØ╝ýä£ HBV ý×¼ÝÖ£ýä▒ÝÖöýØÿ ý£äÝùÿÙÅäÛ░Ç ÙåÆýØÇ Û░äýòö ý╣ÿÙúî ýï£ýùÉÙèö HBsAg ýØîýä▒ýØ┤ÙØ╝ÙÅä BÝÿòÛ░äýù╝ ý×¼ÝÖ£ýä▒ÝÖö ýù¼ÙÂÇýùÉ ÙîÇÝò£ ýú╝ýØÿ Û╣èýØÇ Ù¬¿ÙïêÝä░ÙºüýØ┤ ÝòäýÜöÝòÿÛ▓áÙïñ.
ÛÁ¡Ùé┤ Ùºîýä▒ BÝÿòÛ░äýù╝ ÝÖÿý×ÉÙèö ýáÉý░¿ Û│áÙá╣ÝÖöÙÉÿÛ│á ý×êÙèö ýÂöýä©ýØ┤Ù®░, ýØ┤ýùÉ Ùö░ÙØ╝ ýïáÛ©░ÙèÑ ýáÇÝòÿ, Û│¿ÙîÇýé¼ ýºêÝÖÿ Ùô▒ýØÿ Û░üýóà ÙÅÖÙ░ÿ ýºêÝÖÿýØä Û░ÇýºÇÛ│á ý×êÙèö ÝÖÿý×É ýêÿ ýù¡ýï£ ýªØÛ░ÇÝòÿÛ│á ý×êÙïñ. Ùºîýä▒ BÝÿòÛ░äýù╝ ÝÖÿý×ÉýùÉýä£ Û▓¢ÛÁ¼ Ýò¡Ù░öýØ┤Ùƒ¼ýèñýá£ÙÑ╝ ý▓ÿýØîý£╝Ùí£ ý▓ÿÙ░®ÝòÿÛ▓î ÙÉÿÙèö ý┤êý╣ÿÙúî ÝÖÿý×ÉýØÿ Û▓¢ýÜ░, Ýò┤Ùï╣ ÝÖÿý×ÉýØÿ ýïáÛ©░ÙèÑ ýØ┤ýâü Ù░Å Û│¿ÙîÇýé¼ ýºêÝÖÿ Ýÿ╣ýØÇ ýØ┤Ùƒ¼Ýò£ ýºêÝÖÿýØÿ ý£äÝùÿýä▒(ý×ÑÛ©░Û░ä ýèñÝàîÙí£ýØ┤Ùô£ ýé¼ýÜ®ý×É Ùô▒) ýù¼ÙÂÇÙÑ╝ ýé¼ýáäýùÉ ýé┤ÝÄ┤Ù│┤ýòäýò╝ Ýò£Ùïñ. Ýÿäý×¼ ý┤êý╣ÿÙúî ÝÖÿý×ÉýùÉýä£ 1ý░¿ ý╣ÿÙúîýá£Ùí£ ýÂöý▓£ÙÉÿÛ│á ý×êÙèö ÝàîÙà©ÝżÙ╣äýû┤ DFýØÿ Û▓¢ýÜ░ ÝàîÙà©ÝżÙ╣äýû┤ AFýÖÇýØÿ 3ýâü Ù╣äÛÁÉ ýù░ÛÁ¼ýùÉýä£ ýïáÛ©░ÙèÑ Û░ÉýåîÛ░Ç ý£áýØÿÝòÿÛ▓î ÝàîÙà©ÝżÙ╣äýû┤ DF ýé¼ýÜ®ÛÁ░ýùÉýä£ ÙæÉÙô£Ùƒ¼ýíîý£╝Ù®░, Û│¿Ù░ÇÙÅä ýù¡ýï£ ÝàîÙà©ÝżÙ╣äýû┤ DF ýé¼ýÜ® ÛÁ░ýùÉýä£ ÝàîÙà©ÝżÙ╣äýû┤ AF ýé¼ýÜ®ÛÁ░ýùÉ Ù╣äÝò┤ Û©░ýáÇ ÙîÇÙ╣ä 96ýú╝ ý╣ÿÙúî Ýøä Ùìö ýòàÝÖöÙÉÿýùêÙïñ[40]. Ùö░ÙØ╝ýä£ Û░£ýáòÙÉ£ 2022Ùàä Ùºîýä▒ BÝÿòÛ░äýù╝ ýºäÙúî Û░ÇýØ┤Ùô£ÙØ╝ýØ© Û░£ýáòÝîÉýùÉýä£Ùèö ýïáÛ©░ÙèÑ ýáÇÝòÿýùÉ ÙîÇÝò£ ý£äÝùÿýØ©ý×ÉÛ░Ç ý×êÙèö Û▓¢ýÜ░(Û©░ýáÇ ýé¼ÛÁ¼ý▓┤ýù¼Û│╝ý£¿ < 60 mL/min, Ùï¿Ù░▒Ùç¿, ýòîÙÂÇÙ»╝Ùç¿[ýåîÙ│Ç ýòîÙÂÇÙ»╝: Ýü¼ÙáêýòäÝï░Ùïî Ù╣ä > 30 mg/g], ýáÇýØ©ýé░ÝÿêýªØ[< 2 .5 mg/dL], ýí░ýáêÙÉÿýºÇ ýòèÙèö Ùï╣Ùç¿Ùéÿ Û│áÝÿêýòò), ÝàîÙà©ÝżÙ╣äýû┤ DF ýØ┤ýÖ© ÙïñÙÑ© Û▓¢ÛÁ¼ Ýò¡Ù░öýØ┤Ùƒ¼ýèñýᣠýé¼ýÜ®(ÝàîÙà©ÝżÙ╣äýû┤ AF, ýùöÝàîý╣┤Ù╣äýû┤, Ù▓áýï£ÝżÙ╣äýû┤)ýØä ýÂöý▓£ÝòÿÛ│á ý×êÙïñ. Ù┐ÉÙºî ýòäÙïêÙØ╝ Û│¿Ù░ÇÙÅä Û░ÉýåîýØÿ ý£äÝùÿýØ┤ ý×êÛ▒░Ùéÿ ý×ÑÛ©░ýáüý£╝Ùí£ Û│¿Ù░ÇÙÅäÙÑ╝ Û░Éýåîýï£Ýé¼ ÙºîÝò£ ýò¢ýá£ÙÑ╝ ýé¼ýÜ®ÝòÿÙèö Û▓¢ýÜ░ýùÉÙÅä ÝàîÙà©ÝżÙ╣äýû┤ DF ýØ┤ýÖ© ÙïñÙÑ© Û▓¢ÛÁ¼ Ýò¡Ù░öýØ┤Ùƒ¼ýèñýᣠýé¼ýÜ®ýØä ýÂöý▓£ÝòÿÛ│á ý×êÙïñ(Fig. 2).
ý×ÑÛ©░Û░ä Ýò¡Ù░öýØ┤Ùƒ¼ýèñýá£ÙÑ╝ ýé¼ýÜ®ÝòÿÛ│á ý×êÙèö ÝÖÿý×ÉÛ░Ç ýáÉý░¿ ýªØÛ░ÇÝòÿÛ│á ý×êÛ©░ýùÉ ý╣ÿÙúî ýñæ ýïáÛ©░ÙèÑÛ│╝ Û│¿Ù░ÇÙÅäýØÿ ýºÇýåìýáüýØ© Ù¬¿ÙïêÝä░Û░Ç ÙìöýÜ▒ ýñæýÜöÝò┤ýºÇÛ│á ý×êÙïñ. Ùö░ÙØ╝ýä£ ý╣ÿÙúî ýñæ ýïáÛ©░ÙèÑ Ýÿ╣ýØÇ Û│¿Ù░ÇÙÅä ýáÇÝòÿÛ░Ç Ù░£ýâØÝòá Û▓¢ýÜ░ ý£áÙ░£ýØ©ý×ÉÙÑ╝ ý░¥ýòä ÛÁÉýáòÝòÿÛ│á, ÝòäýÜö ýï£ Ýò¡Ù░öýØ┤Ùƒ¼ýèñýᣠÙ│ÇÛ▓¢ýØä Û│áÙáñÝò┤ýò╝ Ýò£Ùïñ. ÝàîÙà©ÝżÙ╣äýû┤ DF, ÝàîÙà©ÝżÙ╣äýû┤ AFÙÑ╝ Ù╣äÛÁÉÝò£ 3ýâü ýù░ÛÁ¼ýùÉýä£ ÝàîÙà©ÝżÙ╣äýû┤ DFÙÑ╝ 96ýú╝ Ýÿ╣ýØÇ 144ýú╝Û░ä ýé¼ýÜ®ÝòÿÙïñÛ░Ç ÝàîÙà©ÝżÙ╣äýû┤ AFÙí£ Ù│ÇÛ▓¢ÝòÿýÿÇýØä Û▓¢ýÜ░ ýºÇýåìýáüýØ© ýïáÛ©░ÙèÑýØÿ Ýÿ©ýáäýØ┤ Û┤Çý░░ÙÉÿýùêÛ│á, Û│äýåìýáüý£╝Ùí£ ý╣ÿÙúî ýï£ ý▓ÿýØîÙÂÇÝä░ ÝàîÙà©ÝżÙ╣äýû┤ AFÙÑ╝ ýé¼ýÜ®Ýò£ ÝÖÿý×ÉÙôñÛ│╝ ÝåÁÛ│äýáüý£╝Ùí£ ý£áýØÿÝò£ ý░¿ýØ┤ÙÑ╝ Ù│┤ýØ┤ýºÇ ýòèýòÿÙïñ[41]. Ùö░ÙØ╝ýä£ ÝàîÙà©ÝżÙ╣äýû┤ DF ýé¼ýÜ®ýùÉ Ùö░ÙÑ© ýïáÛ©░ÙèÑ Û░ÉýåîÙèö ýò¢ýᣠÙ│ÇÛ▓¢ýØä ÝåÁÝò┤ ÝÜîÙ│ÁÙÉá ýêÿ ý×êÙèö Û░Çýù¡ýáüýØ© Û│╝ýáòý×äýØä ýÂöý©íÝòá ýêÿ ý×êÙïñ. ýØ┤ýáä ýù¼Ùƒ¼ Û░ÇýºÇ ýò¢ýá£ÙÑ╝ ýé¼ýÜ®Ýò£ Û▓¢ÝùÿýØ┤ ý×êÙèö ÙïñýᣠÙé┤ýä▒ BÝÿòÛ░äýù╝ Ù░öýØ┤Ùƒ¼ýèñ Ù│┤ý£á ÝÖÿý×ÉýùÉýä£ÙÅä ý×ÑÛ©░Û░ä ÝàîÙà©ÝżÙ╣äýû┤ DFÙÑ╝ ýé¼ýÜ®Ýò£ ÙÆñ ÝàîÙà©ÝżÙ╣äýû┤ AFÙí£ Ù│ÇÛ▓¢Ýòÿýù¼ ýé¼ýÜ®Ýòá Û▓¢ýÜ░ ýºÇýåìýáüý£╝Ùí£ ÝàîÙà©ÝżÙ╣äýû┤ DFÙÑ╝ ý£áýºÇÝò£ ÛÁ░ýùÉ Ù╣äÝò┤ýä£ ýïáÛ©░ÙèÑÛ│╝ Û│¿Ù░ÇÙÅäýØÿ Û░£ýäáýØä Ù│┤ýù¼ýú╝ýùêÙïñ[42]. Ùö░ÙØ╝ýä£ Ýò¡Ù░öýØ┤Ùƒ¼ýèñýᣠý╣ÿÙúî ýñæ Û┤Çý░░ÙÉá ýêÿ ý×êÙèö ýïáÛ©░ÙèÑ Ýÿ╣ýØÇ Û│¿Ù░ÇÙÅä Û░ÉýåîÙèö ýò¢ýᣠÙ│ÇÛ▓¢ýØä ÝåÁÝò┤ Ýÿ©ýáäÙÉá ýêÿ ý×êýØä Û▓âý£╝Ùí£ ÝîÉÙï¿ÙÉ£Ùïñ.
ýØ┤Ù▓ê Ùºîýä▒ BÝÿòÛ░äýù╝ Û░ÇýØ┤Ùô£ÙØ╝ýØ©ýØÇ ýÂòýáüÙÉ£ ýù░ÛÁ¼ Û▓░Û│╝ÙôñýØä Ù░öÝâòý£╝Ùí£ ý×äýâüýáüý£╝Ùí£ ýñæýÜöÝò£ ÝòÁýï¼ ýú╝ýá£ýùÉ ÙîÇÝò┤ ÙÂÇÙÂä Û░£ýáòÝòÿýù¼ ýá£ýï£ÝòÿýÿÇÙïñ. Ùºîýä▒ BÝÿòÛ░äýù╝ ÝÖÿý×ÉýùÉýä£ ýºäÙúî ýºÇý╣¿ýùÉ Û©░Ù░ÿÝò£ ýáüýáêÝò£ ý╣ÿÙúî Ù░Å Û┤ÇÙª¼ÙÑ╝ ÝåÁÝò┤ Û░äýºêÝÖÿ Û┤ÇÙá¿ Ýò®Ù│æýªØýØä ýÿêÙ░®ÝòÿÛ│á ýâØýí┤ý£¿ýØä ÙìöýÜ▒ ÝûÑýâüýï£Ýé¼ ýêÿ ý×êýØä Û▓âýØ┤Ùïñ.
Notes
REFERENCES
1. Yim SY, Kim JH. The epidemiology of hepatitis B virus infection in Korea. Korean J Intern Med 2019;34:945ÔÇô953.



2. Korean Association for the Study of the Liver (KASL). KASL clinical practice guidelines for management of chronic hepatitis B. Clin Mol Hepatol 2019;25:93ÔÇô159.



3. Korean Association for the Study of the Liver (KASL). KASL clinical practice guidelines for management of chronic hepatitis B. Clin Mol Hepatol 2022;28:276ÔÇô331.



4. Hsu YN, Pan CQ, Abbasi A, Xia V, Bansal R, Hu KQ. Clinical presentation and disease phases of chronic hepatitis B using conventional versus modified ALT criteria in Asian Americans. Dig Dis Sci 2014;59:865ÔÇô871.


5. Yao K, Liu J, Wang J, et al. Distribution and clinical characteristics of patients with chronic hepatitis B virus infection in the grey zone. J Viral Hepat 2021;28:1025ÔÇô1033.


6. Xiao G, Yang J, Yan L. Comparison of diagnostic accuracy of aspartate aminotransferase to platelet ratio index and fibrosis-4 index for detecting liver fibrosis in adult patients with chronic hepatitis B virus infection: a systemic review and meta-analysis. Hepatology 2015;61:292ÔÇô302.


7. Inoue T, Tanaka Y. Novel biomarkers for the management of chronic hepatitis B. Clin Mol Hepatol 2020;26:261ÔÇô279.



8. Loomba R, Adams LA. Advances in non-invasive assessment of hepatic fibrosis. Gut 2020;69:1343ÔÇô1352.


9. Bedossa P, Poynard T. An algorithm for the grading of activity in chronic hepatitis C. The METAVIR Cooperative Study Group. Hepatology 1996;24:289ÔÇô293.


10. Chon YE, Choi EH, Song KJ, et al. Performance of transient elastography for the staging of liver fibrosis in patients with chronic hepatitis B: a meta-analysis. PLoS One 2012;7:e44930.



11. Terrault NA, Lok ASF, McMahon BJ, et al. Update on prevention, diagnosis, and treatment of chronic hepatitis B: AASLD 2018 hepatitis B guidance. Hepatology 2018;67:1560ÔÇô1599.


12. Andreani T, Serfaty L, Mohand D, et al. Chronic hepatitis B virus carriers in the immunotolerant phase of infection: histologic findings and outcome. Clin Gastroenterol Hepatol 2007;5:636ÔÇô641.


13. Kim GA, Lim YS, Han S, et al. High risk of hepatocellular carcinoma and death in patients with immune-tolerant-phase chronic hepatitis B. Gut 2018;67:945ÔÇô952.


14. Kim MN, Kim SU, Kim BK, et al. Increased risk of hepatocellular carcinoma in chronic hepatitis B patients with transient elastography-defined subclinical cirrhosis. Hepatology 2015;61:1851ÔÇô1859.


15. Lok AS, McMahon BJ, Brown RS Jr, et al. Antiviral therapy for chronic hepatitis B viral infection in adults: a systematic review and meta-analysis. Hepatology 2016;63:284ÔÇô306.


16. Chen CJ, Yang HI, Su J, et al. Risk of hepatocellular carcinoma across a biological gradient of serum hepatitis B virus DNA level. JAMA 2006;295:65ÔÇô73.


17. Paik N, Sinn DH, Lee JH, et al. Non-invasive tests for liver disease severity and the hepatocellular carcinoma risk in chronic hepatitis B patients with low-level viremia. Liver Int 2018;38:68ÔÇô75.

18. Sinn DH, Kim SE, Kim BK, Kim JH, Choi MS. The risk of hepatocellular carcinoma among chronic hepatitis B virus-infected patients outside current treatment criteria. J Viral Hepat 2019;26:1465ÔÇô1472.


19. European Association for the Study of the Liver. EASL 2017 clinical practice guidelines on the management of hepatitis B virus infection. J Hepatol 2017;67:370ÔÇô398.


20. Sinn DH, Lee J, Goo J, et al. Hepatocellular carcinoma risk in chronic hepatitis B virus-infected compensated cirrhosis patients with low viral load. Hepatology 2015;62:694ÔÇô701.


21. Cornberg M, Lok AS, Terrault NA; Zoulim F; 2019 EASL-AASLD HBV Treatment Endpoints Conference Faculty. Guidance for design and endpoints of clinical trials in chronic hepatitis B - report from the 2019 EASL-AASLD HBV Treatment Endpoints ConferenceÔÇí. J Hepatol 2020;72:539ÔÇô557.


22. Kim GA, Lim YS, An J, et al. HBsAg seroclearance after nucleoside analogue therapy in patients with chronic hepatitis B: clinical outcomes and durability. Gut 2014;63:1325ÔÇô1332.


23. Choi J, Yoo S, Lim YS. Comparison of long-term clinical outcomes between spontaneous and therapy-induced HBsAg seroclearance. Hepatology 2021;73:2155ÔÇô2166.


24. Park Y, Lee JH, Sinn DH, et al. Risk and risk score performance of hepatocellular carcinoma development in patients with hepatitis B surface antigen seroclearance. Clin Transl Gastroenterol 2021;12:e00290.



25. Coffin CS, Zhou K, Terrault NA. New and old biomarkers for diagnosis and management of chronic hepatitis B virus infection. Gastroenterology 2019;156:355ÔÇô368; e3.


26. Liu J, Li T, Zhang L, Xu A. The role of hepatitis B surface antigen in Nucleos(t)ide analogues cessation among asian patients with chronic hepatitis B: a systematic review. Hepatology 2019;70:1045ÔÇô1055.


27. Jeng WJ, Chen YC, Chien RN, Sheen IS, Liaw YF. Incidence and predictors of hepatitis B surface antigen seroclearance after cessation of nucleos(t)ide analogue therapy in hepatitis B e antigen-negative chronic hepatitis B. Hepatology 2018;68:425ÔÇô434.


28. Mak LY, Wong DK, Cheung KS, Seto WK, Lai CL, Yuen MF. Review article: hepatitis B core-related antigen (HBcrAg): an emerging marker for chronic hepatitis B virus infection. Aliment Pharmacol Ther 2018;47:43ÔÇô54.


29. Drafting Committee for Hepatitis Management Guidelines; the Japan Society of Hepatology. Japan Society of Hepatology guidelines for the management of hepatitis B virus infection: 2019 update. Hepatol Res 2020;50:892ÔÇô923.


30. Wang J, Shen T, Huang X, et al. Serum hepatitis B virus RNA is encapsidated pregenome RNA that may be associated with persistence of viral infection and rebound. J Hepatol 2016;65:700ÔÇô710.


31. Liu S, Zhou B, Valdes JD, Sun J, Guo H. Serum hepatitis B virus RNA: a new potential biomarker for chronic hepatitis B virus infection. Hepatology 2019;69:1816ÔÇô1827.


32. Kaewdech A, Tangkijvanich P, Sripongpun P, et al. Hepatitis B surface antigen, core-related antigen and HBV RNA: predicting clinical relapse after NA therapy discontinuation. Liver Int 2020;40:2961ÔÇô2971.


33. Carey I, Gersch J, Wang B, et al. Pregenomic HBV RNA and hepatitis B core-related antigen predict outcomes in hepatitis B e antigen-negative chronic hepatitis B patients suppressed on Nucleos(T)ide analogue therapy. Hepatology 2020;72:42ÔÇô57.


34. Wu CY, Chen YJ, Ho HJ, et al. Association between nucleoside analogues and risk of hepatitis B virusÔÇôrelated hepatocellular carcinoma recurrence following liver resection. JAMA 2012;308:1906ÔÇô1914.


35. Choi J, Jo C, Lim YS. Tenofovir versus entecavir on recurrence of hepatitis B virus-related hepatocellular carcinoma after surgical resection. Hepatology 2021;73:661ÔÇô673.


36. Tsai MC, Wang CC, Lee WC, et al. Tenofovir is superior to entecavir on tertiary prevention for BCLC Stage 0/A hepatocellular carcinoma after curative resection. Liver Cancer 2021;11:22ÔÇô37.



37. Chang JI, Sinn DH, Cho H, et al. Clinical outcomes of hepatitis B virus-related hepatocellular carcinoma patients with undetectable serum HBV DNA levels. Dig Dis Sci 2022;67:4565ÔÇô4573.


38. Jun BG, Kim YD, Kim SG, et al. Hepatitis B virus reactivation after radiotherapy for hepatocellular carcinoma and efficacy of antiviral treatment: a multicenter study. PLoS One 2018;13:e0201316.



39. Jang JW, Kim YW, Lee SW, et al. Reactivation of hepatitis B virus in HBsAg-negative patients with hepatocellular carcinoma. PLoS One 2015;10:e0122041.



40. Agarwal K, Brunetto M, Seto WK, et al. 96 weeks treatment of tenofovir alafenamide vs. tenofovir disoproxil fumarate for hepatitis B virus infection. J Hepatol 2018;68:672ÔÇô681.


Algorithm for management of chronic hepatitis B virus infection. Reproduced from reference [3] with permission. HBsAg, hepatitis B surface antigen; HBeAg, hepatitis B e antigen; anti-HBe, antibody to hepatitis B e antigen; HBV, hepatitis B virus, ULN, upper limit of normal.
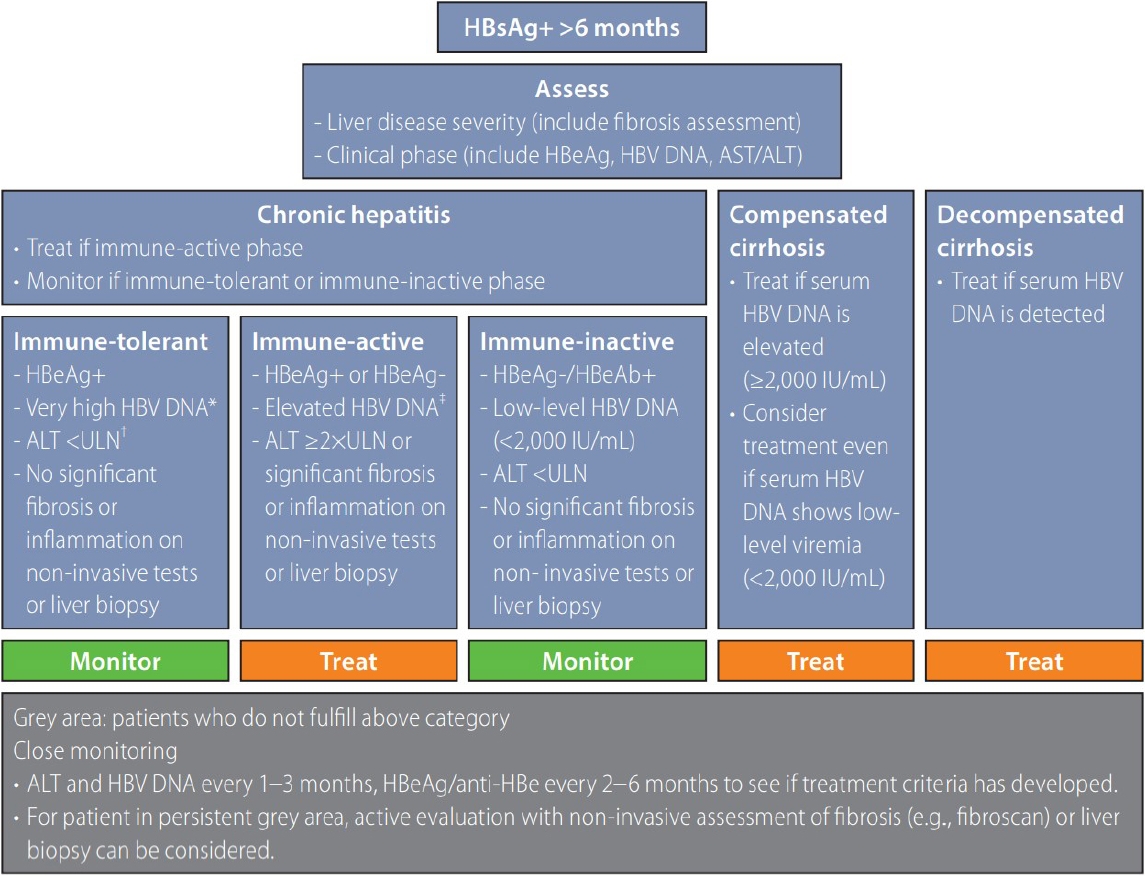
Figure 1.
Indications for selecting entecavir, tenofovir alafenamide fumarate or besifovir over tenofovir disoproxil fumarate. Reproduced from reference [3] with permission. AF, alafenamide fumarate. a Dose was adjusted if creatinine clearance was < 50 mL/min. b Not indicated if creatinine clearance was < 15 mL/min without dialysis. c Not indicated if creatinine clearance was < 15 mL/min.
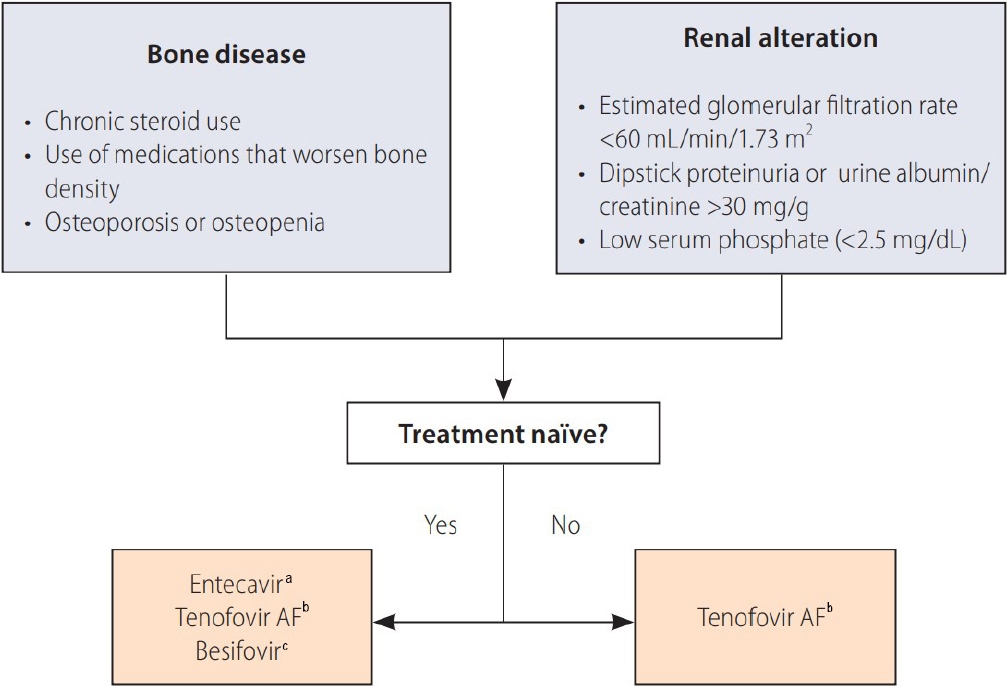
Figure 2.
Table 1.
Natural course of CHB
| Phasesa | Serologic marker | ALT | HBV DNA | Histologic activityb |
|---|---|---|---|---|
| CHB, immune tolerant phase | HBeAg (+) | Persistently normal | Very high levels of viral replication (HBV DNA levels  10,000,000 IU/mL) | None/minimal |
| Anti-HBe (-) | ||||
| HBeAg-positive CHB, immune active phase | HBeAg (+); may develop anti-HBe | Elevated (persistently or intermittently) | High levels of viral replication (HBV DNA levels  20,000 IU/mL) | Moderate/severe |
| CHB, immune inactive phase | HBeAg (-) | Persistently normal | Low or undetectable HBV DNA (HBV DNA levels < 2,000 IU/mL) | Minimal |
| Anti-HBe (+) | ||||
| HBeAg-negative CHB, immune active phase | HBeAg (-) | Elevated (persistently or intermittently) | Moderate to high levels of HBV replication (HBV DNA levels  2,000 IU/mL) | Moderate/severe |
| Anti-HBe (+/-) | ||||
| HBsAg loss phase | HBsAg (-) | Normal | Not detected | - |
| Anti-HBc (+) | ||||
| Anti-HBs (+/-) |



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print




