 |
 |
| Korean J Med > Volume 99(1); 2024 > Article |
|
Abstract
Myelodysplastic syndrome (MDS) is a heterogeneous disorder with diverse prognoses influenced by cytopenias, genetic variants, and myeloblast proportions in the bone marrow. Accurate prognosis prediction and tailored treatment plans are essential. The International Prognostic Scoring System-Molecular (IPSS-M), which additionally reflects the impact of MDS-related genetic mutations to the clinical and laboratory information, is anticipated to offer superior prognostic accuracy compared to existing systems like the Revised International Prognostic Scoring System (IPSS-R). Despite its statistical complexity, its web-based calculation and ease of discussing results with patients using intuitive data sets provide notable advantages. Progress in MDS treatment, exemplified by effective anemia correction with an erythropoiesis-maturation agent in SF3B1-mutated cases and efforts to refine poor prognoses in TP53-mutated cases, reflects the evolving landscape of genetic-based interventions in MDS. Advancements in genetic diagnostic technology, combined with enhanced knowledge of the bone marrow niche, are anticipated to lead to significant improvement in MDS treatment outcomes in the future.
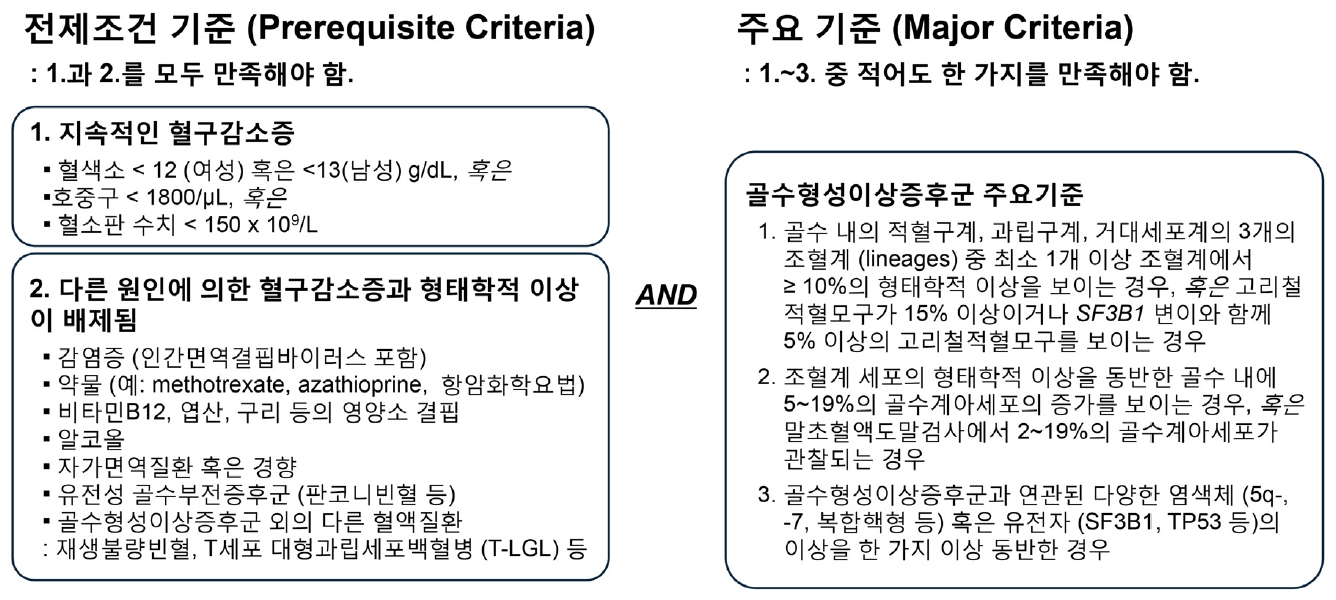
Ļ│©ņłśĒśĢņä▒ņØ┤ņāüņ”ØĒøäĻĄ░(myelodysplastic syndrome, MDS)ņØĆ ņśüņ¢æņåī Ļ▓░ĒĢŹ, Ļ░ÉņŚ╝, ņĢīņĮöņś¼, ņĢĮņĀ£, ņ×ÉĻ░Ćļ®┤ņŚŁņ¦łĒÖś ļō▒ņØś ņØ┤ņ░©ņĀü ņøÉņØĖņØ┤ ņŚåļŖö ĒśłĻĄ¼ Ļ░Éņåī(cytopenia)Ļ░Ć ņ¦ĆņåŹļÉśļ®┤ņä£ Ļ│©ņłś ļé┤ ņĪ░ĒśłĻ│ä ņäĖĒżņØś ĒśĢĒā£ĒĢÖņĀü ņØ┤ņāü, Ļ│©ņłś Ēś╣ņØĆ ļ¦Éņ┤ł ĒśłņĢĪ ļé┤ Ļ│©ņłśĻ│ä ņĢäņäĖĒż(myeloblasts)ņØś ņ”ØĻ░Ć, ļśÉļŖö ĒŖ╣ņ¦ĢņĀüņØĖ ņ£ĀņĀä ļ│ĆņØ┤ ņżæ ĒĢ£ Ļ░Ćņ¦Ć ņØ┤ņāüņØä ļÅÖļ░śĒĢ£ ņāüĒā£ļź╝ ņØ╝ņ╗½ļŖöļŗż(Fig. 1) [1]. ĒśłņĢĪņäĖĒżļōżņØä ņÖäņä▒ļÉśņ¢┤ ņāØņé░ļÉ£ ņĀ£ĒÆłļōż, Ļ│©ņłśļź╝ ĒĢ┤ļŗ╣ ņĀ£ĒÆłļōżņØä ņāØņé░ĒĢśļŖö Ļ│ĄņןņØ┤ļØ╝ ĒĢ£ļŗżļ®┤, MDSļŖö Ļ│ĄņןņØ┤ ņåÉņāüņØä ņ×ģņ¢┤(ņ£ĀņĀä ļ│ĆņØ┤ ļō▒) ņĀ£ĒÆł ņāØņé░ļ¤ēņØ┤ ļīĆņ▓┤ļĪ£ Ļ░ÉņåīĒĢśĻ│Ā(ĒśłĻĄ¼ Ļ░Éņåī), ļ¦īļōżņ¢┤ņ¦ä ņĀ£ĒÆłņØś ņØ╝ļČĆļŖö ļ¬©ņ¢æņØ┤ ņØ┤ņāüĒĢśĻ▒░ļéś ĻĖ░ļŖźņØ┤ ļČłļ¤ēĒĢ£ ņāüĒÖ®(ĒśĢņä▒ ņØ┤ņāü)ņØ┤ļØ╝Ļ│Ā ļ╣äņ£ĀĒĢĀ ņłś ņ׳ļŗż.
ĒÖśņ×ÉļōżņØĆ ļ╣äļĪØ MDSļØ╝ļŖö ĒĢśļéśņØś ņØ┤ļ”äņ£╝ļĪ£ ļ¼ČņŚ¼ ņ׳ņ¦Ćļ¦ī ĒśłĻĄ¼ ņłś Ļ░Éņåī ņĀĢļÅä, ņ£ĀņĀä ļ│ĆņØ┤ ĻĘĖļ”¼Ļ│Ā Ļ│©ņłś ļé┤ Ļ│©ņłśĻ│äņĢäņäĖĒżņØś ļ╣äņ£©ņŚÉ ļö░ļØ╝ ļ¦żņÜ░ ļŗżņ¢æĒĢ£ ņśłĒøäļź╝ ļ│┤ņØĖļŗż. Ļ░£ņĀĢĒīÉ ĻĄŁņĀ£ ņśłĒøä ņ¦ĆĒæ£ ņ▓┤Ļ│ä(Revised International Prognostic Scoring System, IPSS-R)ņŚÉ ļö░ļØ╝ ĒÖśņ×Éļź╝ ļČäļźśĒĢĀ Ļ▓ĮņÜ░, ņ╣śļŻī ņŚåņØ┤ Ļ▓ĮĻ│╝ļź╝ Ļ┤Ćņ░░ĒĢĀ Ļ▓ĮņÜ░ ņśłņāü ņ┤Ø ņāØņĪ┤ņ£©ņØś ņżæņĢÖĻ░ÆņØĆ very low (VL) ņ£äĒŚśĻĄ░ņØś Ļ▓ĮņÜ░ 8.8ļģä ņØ┤ņ¦Ćļ¦ī very high (VH) ņ£äĒŚśĻĄ░ņØĆ 0.8ļģäņ£╝ļĪ£ ļ¦żņÜ░ Ēü░ ņ░©ņØ┤ļź╝ ļ│┤ņØĖļŗż[2]. ņØ┤ļź╝ Ļ│ĀļĀżĒĢĀ ļĢī, ĒÖśņ×ÉņØś ņ£äĒŚśļÅäļź╝ ņĀĢĒÖĢĒĢśĻ▓ī ņśłņĖĪĒĢśĻ│Ā ĻĘĖņŚÉ ļö░ļźĖ ņĀüņĀłĒĢ£ ņ╣śļŻī ļ░®ņ╣©ņØä ņĀĢĒĢśļŖö Ļ▓āņØ┤ ņä▒Ļ│ĄņĀüņØĖ MDS ņ╣śļŻīņØś ĒĢĄņŗ¼ņØ┤ļŗż.
ņśżļ×½ļÅÖņĢł ĻĄŁņĀ£ ņśłĒøä ņ¦ĆĒæ£ ņ▓┤Ļ│ä(International Prognostic Scoring System, IPSS) [3]ņÖĆ IPSS-R [2]ņØ┤ ņśłĒøä ļČäļźś ĻĖ░ņżĆņ£╝ļĪ£ ņé¼ņÜ®ļÉśņ¢┤ ņÖöņ¦Ćļ¦ī ņ£ĀņĀä ļ│ĆņØ┤ ņĖĪļ®┤ņŚÉņä£ļŖö ņŚ╝ņāēņ▓┤ ņØ┤ņāü ņŚ¼ļČĆļ¦īņØä ļ░śņśüĒĢĀ ņłś ņ׳ņŚłļŗż. ĻĘĖļ¤¼ļéś ņ░©ņäĖļīĆ ņŚ╝ĻĖ░ņä£ņŚ┤ ļČäņäØ(next generation sequencing, NGS)ņØä ņØ┤ņÜ®ĒĢśņŚ¼ Ļ│©ņłś Ļ▓Ćņ▓┤ņŚÉņä£ ļŗżņ¢æĒĢ£ ņ£ĀņĀäņ×É ļ│ĆņØ┤ļź╝ ĒĢ£ ļ▓łņŚÉ ĒÖĢņØĖĒĢĀ ņłś ņ׳Ļ▓ī ļÉśļ®┤ņä£, Ļ░ü ņ£ĀņĀäņ×ÉņØś ļ│ĆņØ┤ ņŚ¼ļČĆņÖĆ ņśłĒøäņØś ņŚ░Ļ┤Ćņä▒ņŚÉ ļīĆĒĢ£ ņŚ░ĻĄ¼Ļ░Ć ļ¦ÄņØ┤ ņØ┤ļŻ©ņ¢┤ņĀĖ ņÖöĻ│Ā[4,5], 2022ļģäņŚÉļŖö ļČäņ×É ĻĄŁņĀ£ ņśłĒøä ņ¦ĆĒæ£ ņ▓┤Ļ│ä(IPSS-Molecular, IPSS-M)Ļ░Ć ļō▒ņןĒĢśņśĆļŗż[6].
ņ£ĀņĀäņ×É Ļ▓Ćņé¼ņØś ĒÖĢļīĆļĪ£ MDSņØś ņ╣śļŻīņŚÉļÅä ļ░£ņĀäņØ┤ ņØ┤ļŻ©ņ¢┤ņĪīļŗż. ņŚ╝ņāēņ▓┤ Ļ▓Ćņé¼ļź╝ ĒåĄĒĢśņŚ¼ 5qĻ▓░ņåÉņ”ØĒøäĻĄ░ MDSņŚÉņä£ lenalidomide [7]Ļ░Ć ĒÜ©Ļ│╝ņĀüņØĖ Ļ▓āņ£╝ļĪ£ ņĢīļĀżņ¦ä ņØ┤ĒøäļĪ£, Ļ│Āļ”¼ņ▓ĀņĀüĒśłļ¬©ĻĄ¼(ring sideroblast)ņÖĆ ņŚ░Ļ┤ĆļÉ£ SF3B1 ļ│ĆņØ┤ļź╝ Ļ░Ćņ¦ä ņĀĆņ£äĒŚśĻĄ░ MDS ĒÖśņ×ÉņŚÉĻ▓ī ņĀüĒśłĻĄ¼ņĪ░Ēśłņä▒ņłÖņĀ£ņĀ£(erythropoiesis maturation agent, EMA)ņØĖ luspaterceptņØ┤ ĒÜ©Ļ│╝ņĀüņ×äņØ┤ ĒÖĢņØĖļÉśņŚłļŗż. ļ┐Éļ¦ī ņĢäļŗłļØ╝, ņŚ╝ņāēņ▓┤ļ│ĄĒĢ®ĒĢĄĒśĢ(complex karyotype) ļ░Å ĒÖæņŚ╝ņāēņ▓┤ĒĢĄĒśĢ(monosomal karyotype)Ļ│╝ ĒØöĒ׳ ļÅÖļ░śļÉśņ¢┤ ļ¦żņÜ░ ļČłļ¤ēĒĢ£ ņ╣śļŻī ņä▒ņĀüņØä ļ│┤ņØ┤ļŖö TP53 ļ│ĆņØ┤ MDSņŚÉ ļīĆĒĢ£ ņØ┤ĒĢ┤ļÅä Ļ╣Ŗņ¢┤ņ¦ĆĻ│Ā ņ׳ļŗż.
MDSņØś ņśłĒøä ļČäļźśļź╝ ņ£äĒĢ£ ĻĄŁņĀ£ĒśæļĀźĻĘĖļŻ╣(International Working Group for the Prognosis of MDS, IWG-PM)ņØĆ IPSS-MņØś Ļ░£ļ░£ņØä ņ£äĒĢ┤ ņ£Āļ¤ĮņØä ņżæņŗ¼ņ£╝ļĪ£ ļ»ĖĻĄŁ, ļĖīļØ╝ņ¦ł, ĒāĆņØ┤ņÖäņØś ļŗżņłś ĻĖ░Ļ┤ĆņŚÉņä£ ņ¦äļŗ©ļ░øņØĆ 2,957ļ¬ģņØś MDS ĒÖśņ×Éļź╝ ļ░£ĻĄ┤ ņĮöĒśĖĒŖĖ(discovery cohort)ļĪ£ ņäĀņĀĢĒĢśņŚ¼ ņ×äņāü ņĀĢļ│┤ņÖĆ ņ£ĀņĀäņ×É ņĀĢļ│┤ļź╝ ļČäņäØĒĢśņśĆļŗż[6]. ļ¬©ļōĀ ļČäņäØ Ļ▓Ćņ▓┤ļŖö ņ╣śļŻī ņĀä Ļ▓Ćņ▓┤ļĪ£ Ļ│©ņłś ļé┤ ņĢäņäĖĒż 20% ļ»Ėļ¦īņØ┤ļ®░, ļ¦Éņ┤ł ĒśłņĢĪ ļ░▒ĒśłĻĄ¼ ņłśĻ░Ć 13 ├Ś 109/L ļ»Ėļ¦īņØĖ Ļ▓ĮņÜ░ļĪ£ ņĀ£ĒĢ£ĒĢśņŚ¼ acute myeloid leukemia (AML)ņÖĆ MDS/myeloproliferative neoplasm (MPN) ņżæņ▓®ņ”ØĒøäĻĄ░(MDS/MPN overlap syndrome) ĒÖśņ×ÉļōżņØä ļ░░ņĀ£ĒĢśļĀż ĒĢśņśĆļŗż. ņØ┤ņĀäņØś ĒĢŁņĢö ĒÖöĒĢÖ ņÜöļ▓ĢņØ┤ļéś ļ░®ņé¼ņäĀ ņ╣śļŻī Ēøä ļ░£ņāØĒĢ£ ņ╣śļŻī ņŚ░Ļ┤Ć MDS ĒÖśņ×ÉļŖö ļČäņäØņŚÉ ĒżĒĢ©ĒĢśņśĆļŗż. ļśÉĒĢ£ 754ļ¬ģņØś ņØ╝ļ│Ė MDS ĒÖśņ×ÉļōżņØä Ļ▓Ćņ”Ø ņĮöĒśĖĒŖĖ(validation cohort)ļĪ£ ņČöĒøä ļČäņäØĒĢśņśĆļŗż. ļ¬©ļōĀ ĒÖśņ×ÉļōżņŚÉĻ▓ī ļÅÖņØ╝ĒĢ£ 152Ļ░£ ņ£ĀņĀäņ×ÉņŚÉ ļīĆĒĢ£ Ēæ£ņĀü Ēī©ļäÉ ņŚ╝ĻĖ░ņä£ņŚ┤ ļČäņäØ(targeted panel sequencing)ņØä ņŗ£Ē¢ēĒĢśņśĆļŗż. MLL partial tandem duplicationņØś Ļ▓ĮņÜ░ CNACS in-house coverage analysisļĪ£ Ļ▓ĆņČ£ĒĢ£ Ēøä integrative genomics viewerļĪ£ ņłśļÅÖ Ļ▓Ćņé¼ĒĢśņśĆņ£╝ļ®░ DDX41 ļ│ĆņØ┤ļŖö ļīĆļ”Įņ£ĀņĀäņ×É ļ╣łļÅä(variant allele frequency, VAF)ņÖĆ DDX41 ņóīņØś ņŚ╝ņāēņ▓┤ Ļ▓░ņåÉ ņŚ¼ļČĆļź╝ ĒÖĢņØĖĒĢ£ Ēøä ņ▓┤ņäĖĒż ļ│ĆņØ┤ļź╝ ņāØņŗØņäĖĒż ļ│ĆņØ┤ņÖĆ ĻĄ¼ļČäĒĢśņśĆļŗż[6].
ļČäņäØ Ļ▓░Ļ│╝ļź╝ ĒåĄĒĢ┤ ĒÖśņ×ÉļōżņØä ņ£äĒŚśļÅäņŚÉ ļö░ļØ╝ VL, low (L), moderately low (ML), moderately high (MH), high (H), VHņØś 6Ļ░£ ņ£äĒŚśĻĄ░ņ£╝ļĪ£ ļČäļźśĒĢśņśĆĻ│Ā, ļ╣łļÅäļŖö Ļ░üĻ░ü 14%, 33%, 11%, 11%, 14%, 17%ņśĆļŗż. ņ┤Ø ņāØņĪ┤ ĻĖ░Ļ░äņØś ņżæņĢÖĻ░ÆņØĆ VL ņ£äĒŚśĻĄ░ņŚÉņä£ 10.6ļģäņØ┤ņŚłņ£╝ļ®░, Ļ░Ćņן ļåÆņØĆ ļ╣äņ£©ļĪ£ ņĀäņ▓┤ņØś 1/3ņŚÉ ĒĢ┤ļŗ╣ĒĢśļŖö L ņ£äĒŚśĻĄ░ņŚÉņä£ļŖö 6ļģä, VH ņ£äĒŚśĻĄ░ņŚÉņä£ļŖö 1.0ļģäņØ┤ņŚłļŗż(Table 1) [6]. IPSS-RņŚÉ ļö░ļźĖ ļČäļźś ņŗ£, Ļ│Āņ£äĒŚśĻĄ░ļ│┤ļŗżļŖö ņżæĻ░ä Ēś╣ņØĆ ņĀĆņ£äĒŚśĻĄ░ ĒÖśņ×ÉņŚÉņä£ ņ£äĒŚśļÅäņØś ņ×¼ļČäļźśĻ░Ć ļŹö ļ╣łļ▓łĒĢśĻ▓ī ņØ╝ņ¢┤ļé¼ņ£╝ļ®░, IPSS-MņØś ņĀĢĒÖĢļÅäĻ░Ć ņāüļīĆņĀüņ£╝ļĪ£ ļŹö ļåÆņØĆ Ļ▓āņ£╝ļĪ£ ĒÖĢņØĖļÉśņŚłļŗż. ņśłļź╝ ļōżņ¢┤, IPSS-RļĪ£ ļÅÖņØ╝ĒĢśĻ▓ī intermediate (INT) ņ£äĒŚśĻĄ░ņ£╝ļĪ£ ļČäļźśļÉ£ ĒÖśņ×Éļōż ņżæņŚÉņä£ IPSS-MņØä ņĀüņÜ® ņŗ£ VH ņ£äĒŚśĻĄ░ņ£╝ļĪ£ ņ×¼ļČäļźśļÉ£ ĒÖśņ×ÉļōżņŚÉņä£ņØś ļ¼┤ļ░▒Ēśłļ│æ ļ░£ņāØ ĻĖ░Ļ░ä ņżæņĢÖĻ░ÆņØ┤ 0.87ļģä(95% confidence interval [CI], 0.67-1.30)ņØĖļŹ░ ļ░śĒĢśņŚ¼ IPSS-MņŚÉ ļö░ļØ╝ L ņ£äĒŚśĻĄ░ņ£╝ļĪ£ ņ×¼ļČäļźśļÉ£ Ļ▓ĮņÜ░ ļ¼┤ļ░▒Ēśłļ│æ ļ░£ņāØ ĻĖ░Ļ░ä ņżæņĢÖĻ░ÆņØĆ 6.1ļģä(95% CI, 5.2-10.1)ņ£╝ļĪ£ ĻĘĖ ņ░©ņØ┤Ļ░Ć ņ╗Ėļŗż. ĻĘĖļ¤¼ļéś ļÅÖņØ╝ĒĢ£ IPSS-M ĻĘĖļŻ╣ ļé┤ņŚÉņä£ IPSS-RņŚÉ ļö░ļźĖ ņśłĒøäņØś ņ░©ņØ┤ļŖö ņ£ĀņØśĒĢśĻ▓ī ļéśĒāĆļéśņ¦Ć ņĢŖņĢśļŗż[6]. ņØ┤ļź╝ ĒåĄĒĢ┤ ĻĖ░ņĪ┤ IPSS-RņŚÉņä£ INT ņØ┤ĒĢśņØś ņ£äĒŚśĻĄ░ņŚÉ ĒĢ┤ļŗ╣ĒĢśļŖö ĒÖśņ×ÉļōżņØś Ļ▓ĮņÜ░ NGS Ļ▓Ćņé¼ļź╝ ĒåĄĒĢ£ IPSS-MņØś ĒÖĢņØĖņØ┤ ļŹöņÜ▒ ĒĢäņÜöĒĢĀ ņłś ņ׳ņØīņØä ņĢī ņłś ņ׳ļŗż. ņØ┤ Ļ░ÖņØĆ Ļ▓░Ļ│╝ļŖö Ļ▓Ćņ”Ø ņĮöĒśĖĒŖĖļź╝ ĒåĄĒĢśņŚ¼ ņ×¼ĒśäļÉśņŚłĻ│Ā[6], ņØ┤Ēøä ņŚ¼ļ¤¼ ņŚ░ĻĄ¼ļōżņØ┤ IPSS-MņØś IPSS-R ļīĆļ╣ä ņ£ĀņÜ®ņä▒ņØä ļ│┤Ļ│ĀĒĢśĻ│Ā ņ׳ļŗż[8-11].
IPSS ļ░Å IPSS-RĻ│╝ļŖö ļŗ¼ļ”¼ IPSS-MņØĆ ļ│┤ļŗż ļ¦ÄņØĆ ņ×äņāü ņĀĢļ│┤ņÖĆ ņŚ¼ļ¤¼ ņ£ĀņĀäņ×É Ļ▓Ćņé¼ņØś Ļ▓░Ļ│╝ļōżņØä ļ░śņśüĒĢ┤ņĢ╝ ĒĢśļ»ĆļĪ£ ņēĮĻ▓ī ņĀÉņłśĒÖöĒĢśņŚ¼ ņ£äĒŚśļÅäļź╝ ļČäļźśĒĢĀ ņłś ņŚåļŗż. ļīĆņŗĀ MDS ņ×¼ļŗ©(The MDS Foundation)Ļ│╝ ĒĢ©Ļ╗ś ĒÄĖļ”¼ĒĢ£ ņø╣ Ļ│äņé░ĻĖ░(IPSS-M Risk Calculator; The MDS Foundation, Yardville, NJ, USA, https://mds-risk-model.com)ļź╝ Ļ░£ļ░£ĒĢśņśĆļŗż. ņø╣ Ļ│äņé░ĻĖ░ļź╝ ĒåĄĒĢ┤ ņ¦äļŻī ĒśäņןņŚÉņä£ ņ¦¦ņØĆ ņŗ£Ļ░ä ļé┤ņŚÉ ņĀĢļ│┤ļōżņØä ņ×ģļĀźĒĢĀ ņłś ņ׳Ļ│Ā ņ”ēņŗ£ ņ£äĒŚśļÅä Ļ│äņé░ Ļ▓░Ļ│╝ļź╝ ņ¢╗ņØä ņłś ņ׳ņ£╝ļ®░ Ļ▓░Ļ│╝ņ¦Ćļź╝ PDF ĒīīņØ╝ļĪ£ ņØĖņćäĻ╣īņ¦Ć ĒĢĀ ņłś ņ׳ļŗż. Ļ│äņé░ņØä ĒåĄĒĢśņŚ¼ IPSS-MņŚÉ ļö░ļźĖ ņśłņāü ļ¼┤ļ░▒Ēśłļ│æ ņāØņĪ┤ ĻĖ░Ļ░ä ļ░Å ņ┤Ø ņāØņĪ┤ ĻĖ░Ļ░äņØś ņżæņĢÖĻ░ÆĻ│╝ 25-75% ļ▓öņ£ä, ĻĘĖļ”¼Ļ│Ā 1ļģäĻ│╝ 4ļģä Ļ▓ĮĻ│╝ ņŗ£ ņśłņāüļÉśļŖö AMLļĪ£ņØś ņĀäĒÖśņ£©ņØä ņĢī ņłś ņ׳ļŗż. ņ¦äļŻī ĒśäņןņŚÉņä£ ņŗżņĀ£ ņé¼ņÜ®ĒĢśļŖö ņ£ĀņĀäņ×É Ļ▓Ćņé¼ Ēī©ļäÉņŚÉļŖö IPSS-MņŚÉ ĒżĒĢ©ļÉ£ 152Ļ░£ņØś ņ£ĀņĀäņ×É ņżæ ņØ╝ļČĆĻ░Ć ĒżĒĢ©ļÉśņ¢┤ ņ׳ņ¦Ć ņĢŖņØä ņłś ņ׳ļŖöļŹ░ ņØ┤ Ļ▓ĮņÜ░ not assessedņŚÉ Ēæ£ĻĖ░ĒĢśņŚ¼ ļČäņäØņŚÉ ļ░śņśüĒĢĀ ņłś ņ׳ļŗż. ĒÖśņ×ÉņØś ņ£äĒŚśĻĄ░ņØ┤ ļČäĒżņāü ņ¢┤ļŖÉ ņĀĢļÅäņŚÉ ņ£äņ╣śĒĢśļŖöņ¦ĆņÖĆ ņśłņāüļÉśļŖö ņśłĒøäņŚÉ ļīĆĒĢ£ ņ╣┤Ēöīļ×Ć-ļ¦łņØ┤ņ¢┤ ņāØņĪ┤Ļ│ĪņäĀĻ╣īņ¦Ć ļ│┤ņŚ¼ņŻ╝ļ»ĆļĪ£ ĒÖśņ×ÉļōżņØ┤ Ēśäņ×¼ ņāüĒā£ļź╝ ņŗ£Ļ░üņĀü ņ×ÉļŻīļōżņØä ĒåĄĒĢśņŚ¼ ļ│┤ļŗż ņ¦üĻ┤ĆņĀüņ£╝ļĪ£ ņל ņØ┤ĒĢ┤ĒĢĀ ņłś ņ׳Ļ▓ī ĒĢ┤ņŻ╝ļŖö ņןņĀÉņØ┤ ņ׳ļŗż[12].
NGS Ļ▓Ćņé¼ļź╝ ĒåĄĒĢ┤ ĒĢ£ ļ▓łņŚÉ ļ¦ÄņØĆ ņłśņØś ņ£ĀņĀä ļ│ĆņØ┤ļź╝ Ļ▓Ćņé¼ĒĢĀ ņłś ņ׳Ļ▓ī ļÉśņŚłņ¦Ćļ¦ī, MDSņŚÉņä£ļŖö ņ╣śļŻīņØś Ēæ£ņĀüņ£╝ļĪ£ ņ×æņÜ®ĒĢśĻ▒░ļéś ļÅģĒŖ╣ĒĢ£ ĒŖ╣ņ¦ĢņØä ļ│┤ņŚ¼ ļ│äļÅäņØś ņĢäĒśĢņ£╝ļĪ£ ļČäļźśļÉĀ ņłś ņ׳ļŖö ņ£ĀņĀä ļ│ĆņØ┤Ļ░Ć AMLņØś Ļ▓ĮņÜ░ņÖĆ ļ╣äĻĄÉĒĢĀ ļĢī ļ¦Äņ¦Ć ņĢŖļŗż. 2022ļģä Ļ░£ņĀĢļÉ£ ņäĖĻ│äļ│┤Ļ▒┤ĻĖ░ĻĄ¼(World Health Organization, WHO)ņØś Ļ│©ņłśĻ│ä ņóģņ¢æņŚÉ ļīĆĒĢ£ ņĀ£5ĒīÉ ļČäļźś Ļ░£ņĀĢņĢłņŚÉņä£ļŖö ĻĖ░ņĪ┤ņØś 5ļ▓ł ņŚ╝ņāēņ▓┤ Ļ▓░ņåÉ MDSņŚÉ ļŹöĒĢśņŚ¼ SF3B1 ļ│ĆņØ┤(MDS with low blasts and SF3B1 mutation, MDS-SF3B1)ņÖĆ TP53ņØś ļ│ĆņØ┤ļź╝ ļÅÖļ░śĒĢ£ MDS (MDS with biallelic TP53 inactivation, MDS-biTP53)ļ¦īņØä ļ│äļÅäņØś ņĢäĒśĢņ£╝ļĪ£ ļČäļźśĒĢśĻ│Ā ņ׳ļŗż[1]. Ļ░ÖņØĆ ĒĢ┤ ņāłļĪŁĻ▓ī ļ░£Ēæ£ļÉ£ ĻĄŁņĀ£ ĒĢ®ņØś ļČäļźś(International Consensus Classification, ICC)ņØś Ļ│©ņłśĻ│ä ņóģņ¢æ ļČäļźśņŚÉņä£ļÅä ņÜ®ņ¢┤ņÖĆ ņĀĢņØśņØś ļ»Ėļ¼śĒĢ£ ņ░©ņØ┤ļŖö ņ׳ņ£╝ļéś ļæÉ ņ£ĀņĀäņ×É ļ│ĆņØ┤ļź╝ ļÅÖļ░śĒĢ£ MDSņŚÉ ļīĆĒĢ£ ļČäļźśĻ░Ć ļÅÖņØ╝ĒĢśĻ▓ī ņĀüņÜ®ļÉśņŚłļŗż[13].
ņĀĆņ£äĒŚś MDSņØś Ļ░Ćņן ĒØöĒĢ£ ļ¼ĖņĀ£ļŖö ļ╣łĒśłņØ┤ļŗż. ņāüļŗ╣ ņłśņØś ĒÖśņ×ÉļōżņØ┤ ļ¦ż 8ņŻ╝ ņØ┤ļé┤ņØś Ļ░äĻ▓®ņ£╝ļĪ£ ļ░śļ│ĄņĀüņØĖ ņĀüĒśłĻĄ¼ ņłśĒśłņØ┤ ĒĢäņÜöĒĢ£ ņłśĒśł ņØśņĪ┤ ņāüĒā£ņŚÉ ļ╣Āņ¦äļŗż. ESAļŖö ļ╣łĒśłņØä ĻĄÉņĀĢĒĢśļŖö ļŹ░ ņ£ĀņÜ®ĒĢ£ ņ╣śļŻīļ▓ĢņØ┤ļéś Ēśłņ▓Ł erythropoietinņØ┤ 500 mU/mL ļ»Ėļ¦īņØĖ Ļ▓ĮņÜ░ ņé¼ņÜ®ĒĢĀ ņłś ņ׳ņ£╝ļ®░ ņĢĮ 40-60%ņŚÉņä£ ļ░śņØæņØä ļ│┤ņØĖļŗż. ESAņØś ĒÜ©Ļ│╝ ņ£Āņ¦Ć ĻĖ░Ļ░äņØĆ ĒÅēĻĘĀ 18-24Ļ░£ņøöļĪ£, ļŗżņłśņØś ĒÖśņ×ÉļŖö ESAļź╝ ņé¼ņÜ®ĒĢĀ ņłś ņŚåĻ▒░ļéś ļ░śņØæņØä ņ×āļŖöļŗż[14]. Ļ│©ņłśņŚÉņä£ Ļ│Āļ”¼ņ▓ĀņĀüĒśłļ¬©ĻĄ¼ļØ╝ļŖö ĒŖ╣ņ¦ĢņĀüņØĖ ņĀüĒśłĻĄ¼Ļ│ä ņĪ░ĒśłņäĖĒżĻ░Ć Ļ┤Ćņ░░ļÉśļ®┤ņä£ SF3B1 ņ£ĀņĀäņ×ÉņØś ļ│ĆņØ┤ļź╝ Ļ░Ćņ¦ä ņłśĒśł ņØśņĪ┤ MDS ĒÖśņ×ÉņŚÉņä£ļŖö EMAņØĖ luspaterceptĻ░Ć ļ╣łĒśł ņĪ░ņĀłņŚÉ ĒÜ©Ļ│╝ņĀüņØ┤ļŗż[15]. ļ¦ż 3ņŻ╝ļ¦łļŗż Ēö╝ĒĢś ņŻ╝ņé¼ĒĢśļŖö luspaterceptļŖö ņĀüĒśłĻĄ¼ ņĪ░ĒśłņØś ĒøäĻĖ░ ņä▒ņłÖ ļŗ©Ļ│äņŚÉņä£ transforming growth factor-beta (TGF-╬▓) superfamily ligand trapņ£╝ļĪ£ ņ×æņÜ®ĒĢśņŚ¼ ActRIIB ĒÖ£ņä▒ĒÖö ņ¢ĄņĀ£ņÖĆ Smad2/3 ņŗĀĒśĖ Ļ░Éņåīļź╝ ĒåĄĒĢśņŚ¼ TGF-╬▓ņŚÉ ņØśĒĢ£ ļ¼┤ĒÜ© ņĀüĒśłĻĄ¼ ņĪ░Ēśł(ineffective erythropoiesis)ņØä ņżäņŚ¼ņżīņ£╝ļĪ£ņŹ© Ēśłņāēņåīļź╝ ļåÆņØ┤ļŖö ņĢĮņĀ£ņØ┤ļŗż[16]. ņĀĆņ£äĒŚśĻĄ░ MDS ĒÖśņ×É ņĀäļ░śņØä ļīĆņāüņ£╝ļĪ£ ĒĢ£ 2ņāü ņ×äņāüņŗ£ĒŚś[17]ņŚÉņä£ Ļ│Āļ”¼ņ▓ĀņĀüĒśłļ¬©ĻĄ¼ņÖĆ SF3B1 ļ│ĆņØ┤ļź╝ ļÅÖļ░śĒĢ£ MDS ĒÖśņ×ÉņŚÉĻ▓ī ĒŖ╣Ē׳ ĒÜ©Ļ│╝ņĀüņØ┤ļØ╝ļŖö ļČäņäØņŚÉ ĻĖ░ņ┤łĒĢśņŚ¼ Ļ│Āļ”¼ņ▓ĀņĀüĒśłļ¬©ĻĄ¼Ļ░Ć 15% ņØ┤ņāüņØ┤Ļ▒░ļéś SF3B1 ļÅīņŚ░ļ│ĆņØ┤Ļ░Ć ņ׳ļŖö Ļ▓ĮņÜ░ Ļ│Āļ”¼ņ▓ĀņĀüĒśłļ¬©ĻĄ¼Ļ░Ć 5% ņØ┤ņāüņØĖ ĒÖśņ×ÉĻĄ░ņŚÉņä£ ņ£äņĢĮ ļīĆļ╣ä 3ņāü ņ×äņāüņŗ£ĒŚśņØä ņ¦äĒ¢ēĒĢśņśĆĻ│Ā[15] ĻĘĖ Ļ▓░Ļ│╝ ņ▓½ 24ņŻ╝ ļÅÖņĢł 38%ņØś ĒÖśņ×ÉņŚÉņä£ 8ņŻ╝ ņØ┤ņāü ņ¦ĆņåŹļÉśļŖö ņĀüĒśłĻĄ¼ ņłśĒśł ļ╣äņØśņĪ┤ņØä ļ│┤ņśĆĻ│Ā(ļīĆņĪ░ĻĄ░ 13%, p< 0.001) ļŗżņłśņØś ĒÖśņ×ÉņŚÉņä£ ņØśļ»Ė ņ׳ļŖö ņĀüĒśłĻĄ¼ ņłśĒśłļ¤ē Ļ░Éņåīļź╝ ļ│┤ņśĆļŗż. ņØ┤Ēøä SF3B1 ļŗ©ļ░▒ ĻĖ░ļŖźņØ┤ ņĀĆĒĢśļÉĀ Ļ▓ĮņÜ░ TGF-╬▓ņÖĆ P53 ņŗĀĒśĖņĀäļŗ¼ ņ▓┤Ļ│äĻ░Ć ĒÖ£ņä▒ĒÖöļÉśņ¢┤ G0/G1 ņäĖĒżņŻ╝ĻĖ░ ņżæņ¦Ć(cell cycle arrest)ņÖĆ ļ¼┤ĒÜ© ņĀüĒśłĻĄ¼ ņĪ░ĒśłņØ┤ ņĪ░ņןļÉ©ņØ┤ zebrafishņØä ņØ┤ņÜ®ĒĢ£ ņŗżĒŚśņŚÉņä£ ņ”Øļ¬ģļÉśņŚłļŗż[18]. LuspaterceptņØĆ ĒĢ┤ļŗ╣ ĒÖśņ×ÉĻĄ░ņŚÉņä£ ļ»ĖĻĄŁ Food and Drug AgencyņÖĆ ļīĆĒĢ£ļ»╝ĻĄŁ ņŗØĒÆłņØśņĢĮĒÆłņĢłņĀäņ▓śņØś ņŖ╣ņØĖņØä ņ¢╗ņŚłņ£╝ļ®░, Ēśäņ×¼ļŖö ļŗżņŗ£ Ļ│Āļ”¼ņ▓ĀņĀüĒśłļ¬©ĻĄ¼ļéś SF3B1 ļ│ĆņØ┤ ņŚ¼ļČĆņŚÉ Ļ┤ĆĻ│äņŚåņØ┤ ņłśĒśł ņØśņĪ┤ ņĀĆņ£äĒŚś MDS ĒÖśņ×É ņØ╝ļ░śņŚÉņä£ņØś ļ╣łĒśł Ļ░£ņäĀ Ļ░ĆļŖźņä▒ņØ┤ ESAļź╝ ļīĆņĪ░ĻĄ░ņ£╝ļĪ£ ņłśĒ¢ēļÉ£ 3ņāü ņ×äņāüņŗ£ĒŚś(COMMANDS ņŚ░ĻĄ¼)ņØä ĒåĄĒĢśņŚ¼ ņŚ░ĻĄ¼ ņżæņ£╝ļĪ£ ņĄ£ĻĘ╝ ĻĖŹņĀĢņĀüņØĖ ņżæĻ░ä Ļ▓░Ļ│╝Ļ░Ć ļ░£Ēæ£ļÉśņŚłļŗż[19]. ĻĄŁļé┤ņŚÉņä£ļŖö ņĢäņ¦ü ļČĆļŗ┤ņŖżļ¤¼ņÜ┤ ņĢĮņĀ£ļ╣ä ļō▒ņØä ņØ┤ņ£ĀļĪ£ ĻĖēņŚ¼ ņØĖņĀĢņØä ļ░øņ¦Ć ļ¬╗ĒĢśņŚ¼ ņŗżņĀ£ ņé¼ņÜ®ņØ┤ ņÜ®ņØ┤ĒĢśņ¦ĆļŖö ņĢŖņ£╝ļéś, ļ╣äņłśĒśł ņÜöļ▓ĢņŚÉ ļīĆĒĢ£ ņØśĒĢÖņĀü, ņé¼ĒÜīņĀü ņÜöĻĄ¼Ļ░Ć ņ╗żņĀĖĻ░ĆĻ│Ā ņ׳ļŖö ņāüĒÖ®ņŚÉņä£ ĒāĆ ņĀüņØæņ”Øņ£╝ļĪ£ņØś ĒÖĢļīĆ ļō▒ņØä ĒåĄĒĢ┤ ĻĖĆļĪ£ļ▓ī ņŗ£ņןņØś Ļ┤ĆņĀÉņŚÉņä£ ņżæņÜöĒĢ£ ņĢĮņĀ£ļĪ£ ļČĆņāüĒĢĀ Ļ▓āņ£╝ļĪ£ ņśłņāüļÉ£ļŗż.
TP53 ļ│ĆņØ┤ļź╝ ļÅÖļ░śĒĢ£ Ļ│©ņłśĻ│ä ņóģņ¢æņØś ņśłĒøäĻ░Ć ņóŗņ¦Ć ņĢŖņØĆ Ļ▓āņØĆ ņל ņĢīļĀżņĀĖ ņ׳ņ£╝ļéś, ņĄ£ĻĘ╝ ņ×ÉņäĖĒĢ£ ņŚ░ĻĄ¼ļōżņØä ĒåĄĒĢ┤ TP53 ļ│ĆņØ┤ MDSņÖĆ AMLņŚÉ ļīĆĒĢ£ ņØ┤ĒĢ┤Ļ░Ć Ļ╣Ŗņ¢┤ņ¦ĆĻ│Ā ņ׳ļŗż. ĒśłņĢĪņĢöņŚÉņä£ TP53ņØś ļÅīņŚ░ļ│ĆņØ┤ ņÖĖņŚÉļÅä 17ļ▓ł ņŚ╝ņāēņ▓┤ ļé┤ TP53 ņóīņØś Ļ▓░ņåÉ(deletion)ņØ┤ļéś ņ¢æņ¬Į ņāüļÅÖņŚ╝ņāēņ▓┤ņØś ņ£ĀņĀä ĻĄ¼ņä▒ņØ┤ ļÅÖņØ╝ĒĢśĻ▓ī ļ│ĆĒÖöļÉśņ¢┤ ņāüļÅÖņŚ╝ņāēņ▓┤ Ļ░äņØś ņāüĒśĖ ļ│┤ņÖäņĀü ņŚŁĒĢĀņØä ņåīņŗżņŗ£ĒéżļŖö ņ╣┤Ēö╝ ņłś ņżæļ”Į ņØ┤ĒśĢ ņĀæĒĢ®ņä▒ ņāüņŗż(copy neutral loss of heterozygosity, CN-LOH)ļÅä TP53ņØś ļČłĒÖ£ņä▒ĒÖö(inactivation)ņŚÉ ĻĖ░ņŚ¼ĒĢ£ļŗż. ņØ┤ļ¤░ ĻĖ░ņĀäņ£╝ļĪ£ ļæÉ ņāüļÅÖņŚ╝ņāēņ▓┤ ļ¬©ļæÉņØś TP53ņØ┤ ņ¢ĄņĀ£ļÉśļŖö biallelic Ēś╣ņØĆ double-hitņ£╝ļĪ£ Ēæ£ĒśäļÉśļŖö ņāüĒÖ®ņŚÉņä£ ĒÖśņ×ÉņØś ņśłĒøäĻ░Ć ĒŖ╣Ē׳ ņóŗņ¦Ć ņĢŖļŗżļŖö ņĀÉņØ┤ ņĢīļĀżņĪīĻ│Ā[20] MDS ĒÖśņ×ÉņŚÉņä£ļÅä ņל ĒÖĢņØĖļÉśņŚłļŗż[21]. MDSņŚÉņä£ TP53 ļ│ĆņØ┤ņØś VAFĻ░Ć 22%ļź╝ ņ┤łĻ│╝ĒĢĀ Ļ▓ĮņÜ░ ņŚŁņŗ£ ņóŗņ¦Ć ņĢŖņØĆ ņśłĒøäļź╝ ņŗ£ņé¼ĒĢ£ļŗżļŖö ļČäņäØņØ┤ ņ׳ļŖöļŹ░[21], CN-LOH Ļ▓Ćņé¼Ļ░Ć ņ×äņāü ĒśäņןņŚÉņä£ ņØ╝ņāüņĀüņ£╝ļĪ£ ņØ┤ļŻ©ņ¢┤ņ¦ĆĻĖ░ļŖö ņ¢┤ļĀĄĻĖ░ ļĢīļ¼ĖņŚÉ TP53 ļ│ĆņØ┤ MDS ĒÖśņ×ÉņŚÉņä£ TP53 ļ│ĆņØ┤ņØś VAFļź╝ ņ░ĖĻ│ĀĒĢĀ ņłś ņ׳ņØīņØä ņŗ£ņé¼ĒĢ£ļŗż. ļŗżļ¦ī HOVON-SAKK ĻĘĖļŻ╣ņŚÉņä£ AMLĻ│╝ excess blastsļź╝ ļ│┤ņØ┤ļŖö MDS ĒÖśņ×ÉļōżņØä ļīĆņāüņ£╝ļĪ£ ļČäņäØĒĢ£ ņŚ░ĻĄ¼ņŚÉņä£ļŖö mono-allelicĻ│╝ bi-allelic TP53 ļ│ĆņØ┤ ĒÖśņ×É Ļ░äņØś ņ┤Ø ņāØņĪ┤ņ£©ņØś ņ░©ņØ┤Ļ░Ć ņĀäĒśĆ ņŚåņŚłĻ│Ā, VAFņŚÉ ļö░ļźĖ ņśłĒøäņØś ņ░©ņØ┤ļÅä Ļ┤Ćņ░░ĒĢĀ ņłś ņŚåņŚłĻĖ░ ļĢīļ¼ĖņŚÉ[22] AML ļ░Å ņĢäņäĖĒżĻ░Ć ļåÆņØĆ Ļ│Āņ£äĒŚś MDSņŚÉņä£ļŖö mono-allelic, low VAFņØś TP53 ļ│ĆņØ┤ļØ╝ļÅä ļ│┤ļŗż ĻĖŹņĀĢņĀüņØĖ Ļ▓░Ļ│╝ļź╝ ĻĖ░ļīĆĒĢĀ ņłś ņ׳ļŗżĻ│Ā ļŗ©ņĀĢĒĢśĻĖ░ļŖö ņ¢┤ļĀĄļŗż. TP53 ļ│ĆņØ┤ Ļ│©ņłśĻ│ä ņóģņ¢æņØś ņ╣śļŻīļŖö Ļ│©ņłśĻ│ä ņóģņ¢æņŚÉņä£ Ļ░Ćņן ļÅäņĀäņĀüņØĖ ļČäņĢ╝ļĪ£, ņ¦łļ│æņØś ĻĖ░ņĀäņŚÉ ļīĆĒĢ£ ņØ┤ĒĢ┤Ļ░Ć ļåÆņĢäņ¦Ćļ®┤ņä£ ņāłļĪŁĻ│Ā ĒÜ©Ļ│╝ņĀüņØĖ ņ╣śļŻīļ▓ĢņØ┤ Ļ░£ļ░£ļÉĀ Ļ▓āņ£╝ļĪ£ ĻĖ░ļīĆļÉ£ļŗż.
Ļ│Āņ£äĒŚś MDS ĒÖśņ×ÉĻ░Ć Ļ│ĀĻ░ĢļÅä ņ╣śļŻīļź╝ Ļ▓¼ļö£ ņłś ņ׳ļŖö ņŗĀņ▓┤ ņāüĒā£ļź╝ Ļ░¢ņČöņŚłļŗżļ®┤, ņÖäņ╣śļź╝ ņ£äĒĢśņŚ¼ ļÅÖņóģņĪ░Ēśłļ¬©ņäĖĒż ņØ┤ņŗØņØä ņŗ£ļÅäĒĢĀ ņłś ņ׳ļŗż. ļīĆņ▓┤ļĪ£ Ļ│©ņłś ļé┤ ņĢäņäĖĒż ļ╣äņ£©ņØ┤ ļåÆņ£╝ļ®┤ ņäĀĒ¢ē ņ╣śļŻīļĪ£ ņĢäņäĖĒż ļ╣äņ£©ņØä ļé«ņČöĻ│Ā ņØ┤ņŗØņØä ņ¦äĒ¢ēĒĢśļŖö Ļ▓āņØ┤ ĻČīĻ│ĀļÉśļéś[23], ņäĀĒ¢ē ņ╣śļŻīņØĖ ņĀĆļ®öĒŗĖĒÖöņĀ£ļéś ņ¦æņżæ Ļ┤ĆĒĢ┤ ņ£ĀļÅä ĒĢŁņĢö ĒÖöĒĢÖ ņÜöļ▓ĢņØś ņ╣śļŻī ļ░śņØæņØä ļČäļ¬ģĒ׳ ņśłņĖĪĒĢĀ ņłś ņ׳ļŖö ņ£ĀņĀä ļ│ĆņØ┤ļŖö ņĢäņ¦üĻ╣īņ¦Ć ņĢīļĀżņ¦ä ļ░ö ņŚåļŗż[5].
IPSS, IPSS-R, IPSS-MņØĆ Ļ░üĻ░ü 4Ļ░£, 5Ļ░£, 6Ļ░£ņØś ĻĘĖļŻ╣ņ£╝ļĪ£ ņ£äĒŚśĻĄ░ņØä ļČäļźśĒĢśļéś, ļÅÖņóģ ņĪ░Ēśłļ¬©ņäĖĒż ņØ┤ņŗØ ņŗ£Ē¢ē ņŚ¼ļČĆļź╝ ĒīÉļŗ©ĒĢ┤ņĢ╝ ĒĢśļŖö ņ×äņāüņØśņØś ņ×ģņןņŚÉņä£ļŖö Ļ▓░ĻĄŁ ņĀĆņ£äĒŚśĻĄ░Ļ│╝ Ļ│Āņ£äĒŚśĻĄ░ Ļ░äņØś ļČäļźśĻ░Ć ĒĢĄņŗ¼ņØ┤ļ®░, ĻĘĖļ¤░ ļ®┤ņŚÉņä£ 5Ļ░£ņØś ĻĘĖļŻ╣ņ£╝ļĪ£ ļČäļźśĒĢśĻ│Ā Ļ░ĆņÜ┤ļŹ░ INT ņ£äĒŚśĻĄ░ņØä ļæö IPSS-Rļ│┤ļŗżļŖö Ļ░üĻ░ü 3Ļ░£ņö®ņØś ņĀĆņ£äĒŚśĻĄ░Ļ│╝ Ļ│Āņ£äĒŚśĻĄ░ņØä ļæö IPSS-MņØ┤ ĒÄĖļ”¼ĒĢĀ ņłś ņ׳ļŗż. IPSS-MņØ┤ IPSS-Rļ│┤ļŗż ņ┤Ø ņāØņĪ┤ ĻĖ░Ļ░äĻ│╝ AMLļĪ£ņØś ņĀäĒÖś ņ£äĒŚśņØä ļ│┤ļŗż ņל ņśłņĖĪĒĢ£ļŗżļŖö ņĖĪļ®┤ņŚÉņä£, ņĢ×ņ£╝ļĪ£ IPSS-M Ļ▓░Ļ│╝Ļ░Ć ĒÖśņ×ÉņØś ņØ┤ņŗØ Ļ▓░ņĀĢņŚÉ ņ׳ņ¢┤ Ļ░Ćņן ņżæņÜöĒĢ£ ņÜöņåīļĪ£ Ļ│ĀļĀżļÉĀ ņłś ņ׳ļŗż. ļÅÖņóģ ņĪ░Ēśłļ¬©ņäĖĒż ņØ┤ņŗØ Ļ▓░ņĀĢ Ļ│╝ņĀĢņŚÉņä£ IPSS-MņØś ņÜ░ņøöĒĢ£ ņ£ĀņÜ®ņä▒ņŚÉ ļīĆĒĢśņŚ¼ ļ│┤Ļ│ĀĒĢśļŖö ņŚ░ĻĄ¼ļÅä ļ░£Ēæ£ļÉśĻ│Ā ņ׳ļŗż[10]. ļŗżļ¦ī ļō▒ņןĒĢ£ņ¦Ć ņ¢╝ļ¦ł ļÉśņ¦Ć ņĢŖņØĆ IPSS-MņØ┤ Ļ│╝ņŚ░ ņ£ĀņĀäņ×É Ļ▓Ćņé¼ Ļ▓░Ļ│╝ļź╝ ļ░śņśüĒĢ£ ņĄ£Ļ│ĀņØś ņśłĒøä ņśłņĖĪ ļÅäĻĄ¼ņØĖņ¦ĆņŚÉ ļīĆĒĢ┤ņä£ļŖö ļŹö ļ¦ÄņØĆ Ļ▓Ćņ”Ø ņŚ░ĻĄ¼Ļ░Ć ĒĢäņÜöĒĢśļŗż. ņĀĆņ£äĒŚśĻĄ░ņŚÉņä£ ņŗ£Ļ░äņØä ļæÉĻ│Ā Ļ│Āņ£äĒŚśĻĄ░ņ£╝ļĪ£ ņ¦äĒ¢ēĒĢ£ MDS ĒÖśņ×ÉņØś Ļ▓ĮņÜ░ Ēü┤ļĪĀ ņ¦äĒÖö(clonal evolution)ņØś Ļ░ĆļŖźņä▒ņØ┤ ļåÆņĢä[24] ņØ┤ņŗØ ņĀä ņāłļĪŁĻ▓ī NGS Ļ▓Ćņé¼ļź╝ ĒĢ┤ņĢ╝ ĒĢĀ ņłś ņ׳ļŗż. Ēśäņ×¼ ĻĄŁļé┤ņØś ņĪ░Ēśłļ¬©ņäĖĒż ņØ┤ņŗØ ņÜöņ¢æ ĻĖēņŚ¼ ļīĆņāüņ×É ņ¦łļ│æļ│ä ĻĖ░ņżĆņŚÉņä£ļŖö IPSS-MņØ┤ MDS ĒÖśņ×ÉņŚÉņä£ņØś Ļ│ĀļĀż ņé¼ĒĢŁņ£╝ļĪ£ ļ¬ģņŗ£ļÉśņ¢┤ ņ׳ņ¦Ć ņĢŖļŗż.
MDSļŖö ĒśłĻĄ¼ Ļ░Éņåī, ņ£ĀņĀä ļ│ĆņØ┤ ĻĘĖļ”¼Ļ│Ā Ļ│©ņłś ļé┤ ņĢäņäĖĒżņØś ļ╣äņ£©ņŚÉ ļö░ļØ╝ ļ¦żņÜ░ ļŗżņ¢æĒĢ£ ņśłĒøäļź╝ ļ│┤ņØ┤ļŖö ņØ┤ņ¦łņĀüņØĖ ņ¦łĒÖśņØ┤ļ»ĆļĪ£, Ļ░£ļ│ä ĒÖśņ×ÉņØś ņśłĒøäļź╝ ņĀĢĻĄÉĒĢśĻ▓ī ņśłņĖĪĒĢśĻ│Ā ĻĘĖņŚÉ ļö░ļźĖ ņĀüņĀłĒĢ£ ņ╣śļŻī Ļ│äĒÜŹņØä ņłśļ”ĮĒĢ┤ņĢ╝ ĒĢ£ļŗż. ĻĖ░ņĪ┤ņØś ņ×äņāü ļ░Å ņŗżĒŚśņŗż ņĀĢļ│┤ņÖĆ ĒĢ©Ļ╗ś MDS Ļ┤ĆļĀ© ņ£ĀņĀäņ×É ļ│ĆņØ┤ļōżņØś ņśüĒ¢źņØä ņóģĒĢ®ņĀüņ£╝ļĪ£ ļ░śņśüĒĢ£ IPSS-MņØĆ IPSS-R ļō▒ ĻĖ░ņĪ┤ņØś ņśłĒøä ļČäļźś ņ¦ĆĒæ£ļōżļ│┤ļŗż ļŹö ļéśņØĆ ņśłĒøä ņśłņĖĪņØ┤ Ļ░ĆļŖźĒĢśĻ▓ī ĒĢĀ Ļ▓āņ£╝ļĪ£ ĻĖ░ļīĆļÉśļ®░, ļé┤ļČĆ ĒåĄĻ│äņĀüņØĖ ļ│Ąņ×Īņä▒ņŚÉļÅä ļČłĻĄ¼ĒĢśĻ│Ā ņø╣ Ļ│äņé░ĻĖ░ļź╝ ĒåĄĒĢ┤ ĒÄĖļ”¼ĒĢśĻ▓ī Ļ│äņé░ĒĢśĻ│Ā ĒÖśņ×ÉņÖĆ Ļ▓░Ļ│╝ļź╝ ņāüņØśĒĢĀ ņłś ņ׳ļŖö ņןņĀÉņØ┤ ņ׳ļŗż. SF3B1 ļ│ĆņØ┤ ļÅÖļ░ś MDSņŚÉņä£ ĒŖ╣ļ│äĒ׳ ņóŗņØĆ ļ░śņØæņØä ļ│┤ņØ┤ļŖö ļ╣łĒśł ĻĄÉņĀĢ ņĀ£ņĀ£ņØś ļō▒ņןĻ│╝ TP53 ļ│ĆņØ┤ļź╝ ļÅÖļ░śĒĢ£ MDSņØś ļČłļ¤ēĒĢ£ ņśłĒøäļź╝ ņäĖļČäĒÖöĒĢśļĀżļŖö ļģĖļĀź ļō▒ņØĆ ņ£ĀņĀäņ×É Ļ▓Ćņé¼ņØś ļ░£ņĀäņØ┤ MDS ņ╣śļŻīņØś ņ¦äļ│┤ļĪ£ ņØ┤ņ¢┤ņ¦ĆĻ│Ā ņ׳ņØīņØä ļ│┤ņŚ¼ņŻ╝Ļ│Ā ņ׳ļŗż. NGSĻ░Ć ņØ╝ņāüĒÖöļÉ£ ņĀĢļ░Ć ņ£ĀņĀäņ×É Ļ▓Ćņé¼ ņŗ£ļīĆņØś ņ¦äļŗ© ĻĖ░ņłĀ ļ░£ņĀäņØ┤ ļ│æĒā£ņāØļ”¼ ņŚ░ĻĄ¼ņØś ļŗżļźĖ Ēü░ ĒĢ£ ņČĢņØĖ Ļ│©ņłś ļé┤ ļ»ĖņäĖĒÖśĻ▓Į[25]ņŚÉ ļīĆĒĢ£ ņ¦ĆņŗØņØś Ē¢źņāüĻ│╝ ĒĢ®ņ│ÉņĀĖņä£ ņĢ×ņ£╝ļĪ£ MDS ņ╣śļŻī ņä▒ņĀüņØś Ēü░ Ē¢źņāüņØä ņØ┤ļŻ░ Ļ▓āņ£╝ļĪ£ ĻĖ░ļīĆļÉ£ļŗż.
REFERENCES
1. Khoury JD, Solary E, Abla O, et al. The 5th edition of the World Health Organization classification of haematolymphoid tumours: myeloid and histiocytic/dendritic neoplasms. Leukemia 2022;36:1703ŌĆō1719.


2. Greenberg PL, Tuechler H, Schanz J, et al. Revised international prognostic scoring system for myelodysplastic syndromes. Blood 2012;120:2454ŌĆō2465.


3. Greenberg P, Cox C, LeBeau MM, et al. International scoring system for evaluating prognosis in myelodysplastic syndromes. Blood 1997;89:2079ŌĆō2088.


4. Haferlach T, Nagata Y, Grossmann V, et al. Landscape of genetic lesions in 944 patients with myelodysplastic syndromes. Leukemia 2014;28:241ŌĆō247.


6. Bernard E, Tuechler H, Greenberg PL, et al. Molecular international prognostic scoring system for myelodysplastic syndromes. NEJM Evid 2022;1:2200008.
7. List A, Dewald G, Bennett J, et al. Lenalidomide in the myelodysplastic syndrome with chromosome 5q deletion. N Engl J Med 2006;355:1456ŌĆō1465.


8. Aguirre LE, Al Ali N, Sallman DA, et al. Assessment and validation of the molecular international prognostic scoring system for myelodysplastic syndromes. Leukemia 2023;37:1530ŌĆō1539.


9. Kewan T, Bahaj W, Durmaz A, et al. Validation of the molecular international prognostic scoring system in patients with myelodysplastic syndromes. Blood 2023;141:1768ŌĆō1772.


10. Sauta E, Robin M, Bersanelli M, et al. Real-world validation of molecular international prognostic scoring system for myelodysplastic syndromes. J Clin Oncol 2023;41:2827ŌĆō2842.


11. Yang T, Jiang B, Luo Y, et al. Comparison of the prognostic predictive value of molecular international prognostic scoring system and revised international prognostic scoring system in patients undergoing allogeneic hematopoietic stem cell transplantation for myelodysplastic neoplasms. Am J Hematol 2023;98:E391ŌĆōE394.


12. Bejar R. How can we incorporate molecular data into the IPSS? Best Pract Res Clin Haematol 2022;35:101410.


13. Arber DA, Orazi A, Hasserjian RP, et al. International consensus classification of myeloid neoplasms and acute leukemias: integrating morphologic, clinical, and genomic data. Blood 2022;140:1200ŌĆō1228.


14. Carraway HE, Saygin C. Therapy for lower-risk MDS. Hematology Am Soc Hematol Educ Program 2020;2020:426ŌĆō433.



15. Fenaux P, Platzbecker U, Mufti GJ, et al. Luspatercept in patients with lower-risk myelodysplastic syndromes. N Engl J Med 2020;382:140ŌĆō151.

16. Suragani RN, Cawley SM, Li R, et al. Modified activin receptor IIB ligand trap mitigates ineffective erythropoiesis and disease complications in murine ╬▓-thalassemia. Blood 2014;123:3864ŌĆō3872.



17. Platzbecker U, G├Čtze KS, Kiewe P, et al. Long-term efficacy and safety of luspatercept for anemia treatment in patients with lower-risk myelodysplastic syndromes: the phase II PACE-MDS study. J Clin Oncol 2022;40:3800ŌĆō3807.



18. De La Garza A, Cameron RC, Gupta V, Fraint E, Nik S, Bowman TV. The splicing factor Sf3b1 regulates erythroid maturation and proliferation via TGF╬▓ signaling in zebrafish. Blood Adv 2019;3:2093ŌĆō2104.



19. Platzbecker U, Della Porta MG, Santini V, et al. Efficacy and safety of luspatercept versus epoetin alfa in erythropoiesis-stimulating agent-naive, transfusion-dependent, lowerrisk myelodysplastic syndromes (COMMANDS): interim analysis of a phase 3, open-label, randomised controlled trial. Lancet 2023;402:373ŌĆō385.


20. Campo E, Cymbalista F, Ghia P, et al. TP53 aberrations in chronic lymphocytic leukemia: an overview of the clinical implications of improved diagnostics. Haematologica 2018;103:1956ŌĆō1968.



21. Bernard E, Nannya Y, Hasserjian RP, et al. Implications of TP53 allelic state for genome stability, clinical presentation and outcomes in myelodysplastic syndromes. Nat Med 2020;26:1549ŌĆō1556.


22. Grob T, Al Hinai ASA, Sanders MA, et al. Molecular characterization of mutant TP53 acute myeloid leukemia and high-risk myelodysplastic syndrome. Blood 2022;139:2347ŌĆō2354.


23. de Witte T, Bowen D, Robin M, et al. Allogeneic hematopoietic stem cell transplantation for MDS and CMML: recommendations from an international expert panel. Blood 2017;129:1753ŌĆō1762.



Table┬Ā1.
Prognostication of myelodysplastic syndrome according to the molecular international prognostic scoring system (IPSS-M) [6]



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print




