INTRODUCTION
Thrombotic thrombocytopenic purpura (TTP) is a lethal condition characterized by microangiopathic hemolytic anemia (MAHA), thrombocytopenia, renal insufficiency, neurological abnormalities, and fever [1]. The incidence of acute heart failure associated with TTP is reported to be 9.5% and is considered a poor prognostic factor [2]. The vast majority of heart failures in patients with TTP are associated with the left side of the heart, with few reports of right ventricular heart failure (RVHF). Here, we present a case of TTP with RVHF following total knee replacement (TKR) surgery.
CASE REPORT
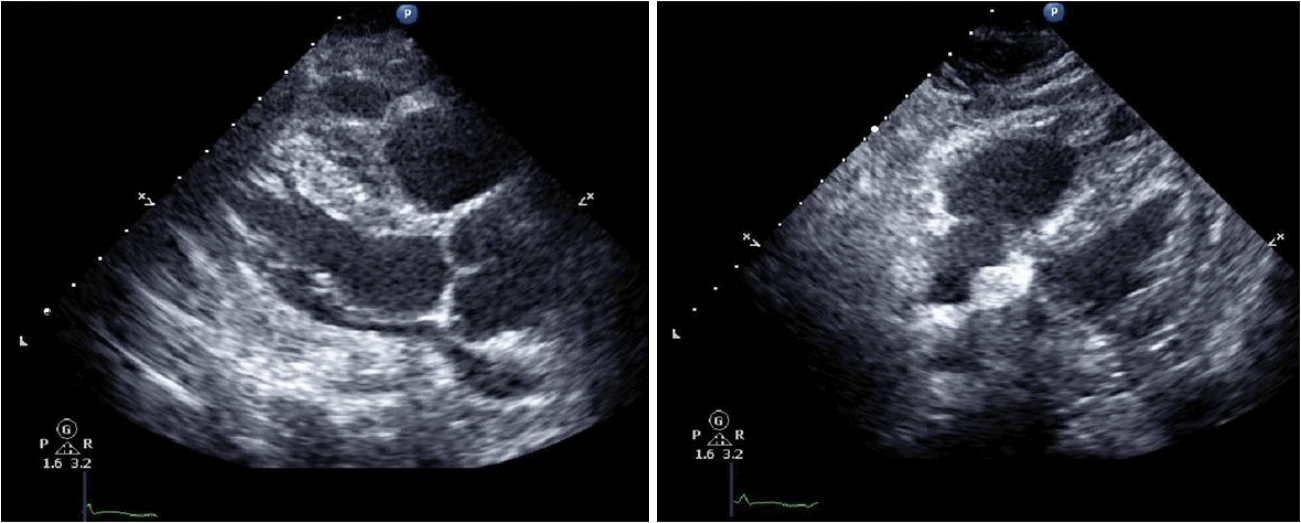
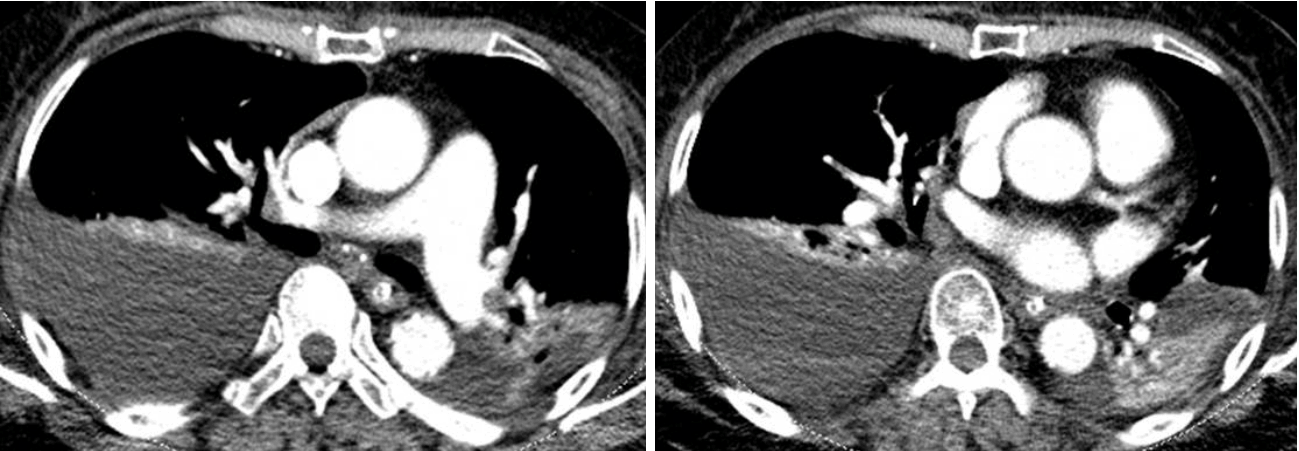
An 80-year-old woman presented at our emergency department in a stuporous condition. The patient had undergone TKR at a different hospital on the previous day but began exhibiting significant changes in mental status within 24 hours after the surgery. The patient had no history of cardiac disease, but she did suffer from hypertension. The patient had been taking atenolol, felodipine, and domperidone prior to the operation. Examination in the emergency department revealed the following: blood pressure, 65/39 mmHg; pulse rate, 98 beats per minute; respiratory rate, 30 breaths per minute; and body temperature, 35.0┬░C. Arterial blood gas analysis performed while she was breathing oxygen (15 L/min) through a reservoir mask revealed the following: pH, 7.350; PaCO2, 33.0 mmHg; PaO2, 48.0 mmHg; HCO3, 18.0 mmol/L; and O2 saturation, 83.4%. Crackles were heard in both lung fields. The wound of left knee was clear, with no erythema, swelling, discharge, bleeding, or dehiscence. She underwent endotracheal intubation, was placed on mechanical ventilation, and was treated with a large volume of crystalloid, high-dose vasopressors, and inotropics. Initial laboratory findings revealed a white blood cell count of 17,600/mm3, a hemoglobin level of 15.0 g/dL, and a platelet count of 128,000/mm3. Blood coagulation tests (prothrombin time [PT] and activated partial prothrombin time [aPTT]) were normal. The blood urea nitrogen level was 34 mg/dL and the serum level of creatinine was 2.0 mg/mL. The levels of lactate dehydrogenase and total bilirubin were 447 IU/L and 2.1 mg/dL, respectively. Cardiac enzymes were mildly elevated: CK-MB, 8.7 g/mL, and troponin-I, 0.637 ╬╝g/mL. Brain levels of natriuretic peptides were 1,444 pg/mL and the lactate level was 4.0 mmol/L. Chest radiography indicated bilateral pulmonary edema (Fig. 1). Electrocardiogram results were normal (Fig. 2). Echocardiography revealed normal left ventricular function; however, the right ventricle was dilated and showed reduced contractility. Left ventricular ejection fraction was 56%, with left ventricular dimensions of 33 mm at diastole and 23 mm at systole; the left atrial dimension was 19 mm. The estimated pulmonary artery systolic pressure was 39 mmHg (Fig. 3); however, dynamic chest computed tomographic angiography showed no evidence of pulmonary embolism (Fig. 4). The patient was in an anuric state from the first day of admission. On day 3 of hospital admission, hemoglobin levels fell to 8.6 g/dL and the platelet count fell to 20,000/mm3, with no associated increases in PT or aPTT. No microorganisms were detected in cultures from blood, urine, sputum, and wound. A peripheral blood smear contained schistocytes. Based on these results, the patient was diagnosed with postoperative TTP following TKR surgery, with evidence of MAHA, thrombocytopenia, renal failure, and altered mental status. The patient underwent four rounds of plasma exchange, after which she showed no signs of TTP. She was discharged after 11 weeks.
DISCUSSION
TTP, initially described by Moschcowitz in 1924, is a life-threatening disease [1]. The mechanism underlying acquired TTP involves the formation of unusually large multimers of von Willebrand factor due to a severe reduction in the level of disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13 (ADAMTS13) [3]. In most patients with acquired TTP, the level of plasma ADAMTS13 activity is < 5% of normal values [4]. We did not confirm the level of ADAMTS13 activity in the patient reported herein, which could be seen as a limitation; however, the patient showed many of the classic features of TTP, which makes a misdiagnosis unlikely.
The development of TTP after orthopedic surgery is not common [5-7]. Among cases of TTP that do develop after orthopedic surgery, the case reported here is unique because it was accompanied by RVHF. The reported incidence of cardiac involvement in TTP is 9.5%, with a mortality rate of 38% [2]. However, only left ventricular heart failure has been reported. The patient described herein had RVHF, with normal left ventricular systolic function. Treatment of TTP resulted in an improvement in right ventricular function, which suggests that RVHF may be associated with TTP, particularly after TKR surgery.
As the initial presentation was dyspnea due to pulmonary edema, there is a possibility of transient left ventricular dysfunction associated with stress-induced cardiomyopathy. In cases of right ventricular involvement in stress-induced cardiomyopathy, these symptoms are typically accompanied by lower left ventricular ejection fraction and higher incidence of pulmonary edema [8]. Here, we observed only right ventricular dysfunction at the time of admission, with insufficient time for left ventricular function to have recovered if a bilateral failure were to have occurred. Alternatively, cardiac involvement of TTP has been shown to manifest as arrhythmia in 15% of cases [9], which suggests that a brief arrhythmia may have caused elevated left atrial end-diastolic pressure to induce pulmonary edema.
In this patient, we treated TTP using plasma exchange. Previous studies have recommended daily plasma exchange in cases of acquired acute idiopathic TTP [10]. Plasma exchange removes von Willebrand factor multimers and autoantibodies against ADAMTS13 and provides additional metalloprotease enzymes. In this case, plasma exchange led to a full recovery, which indicates that plasma exchange is a useful therapy for patients with TTP involving right ventricular failure, as well as for those with more traditional TTP symptoms. Taken together, the data presented here suggest that while RVHF is a rare complication in patients with TTP, the condition can be treated effectively with plasma exchange.




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print






